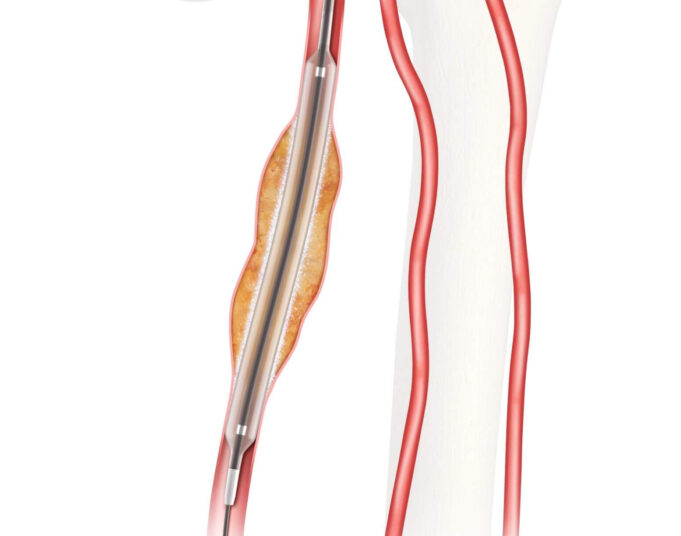
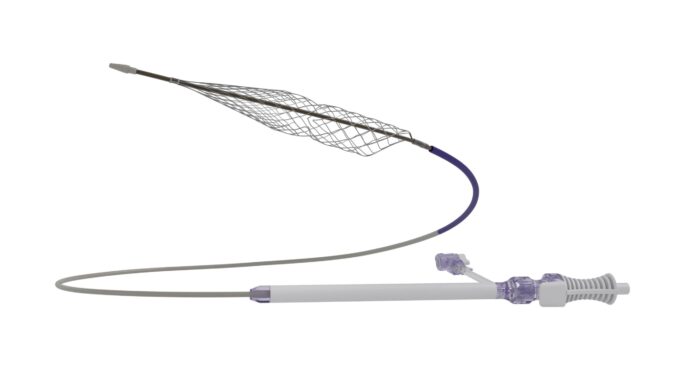
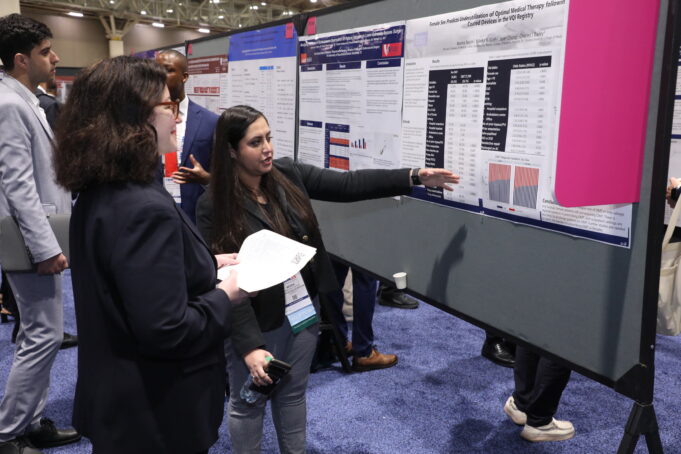
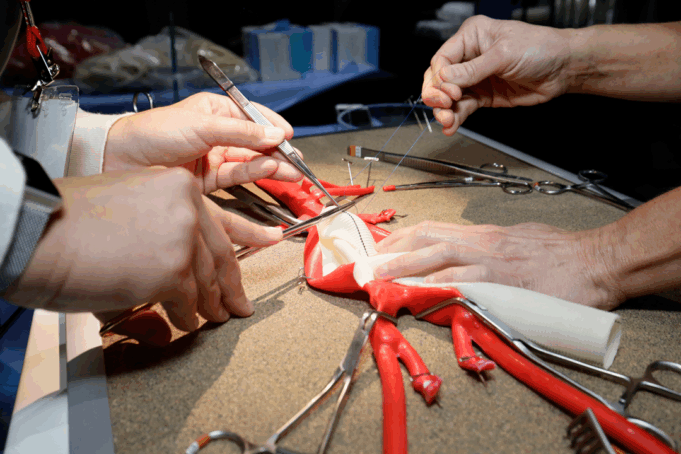
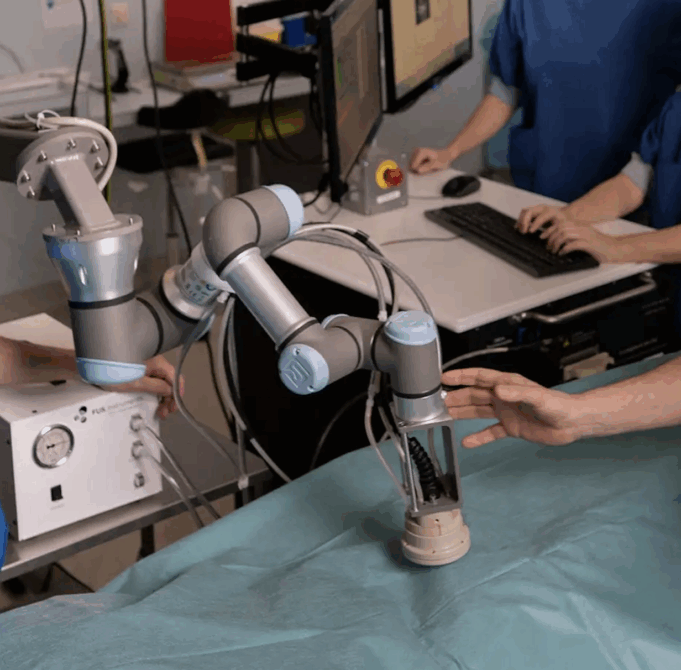
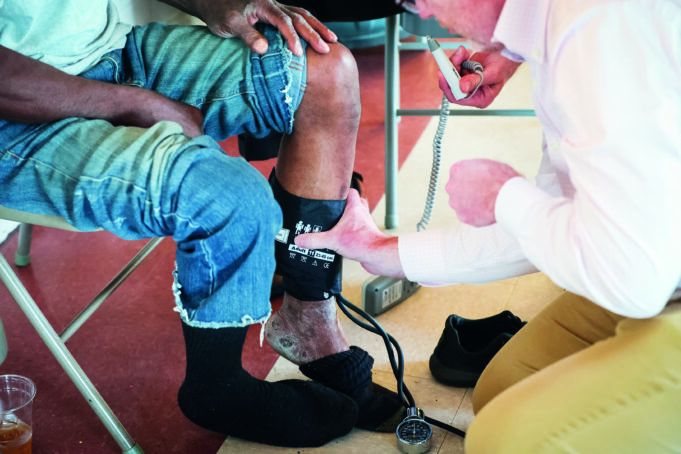
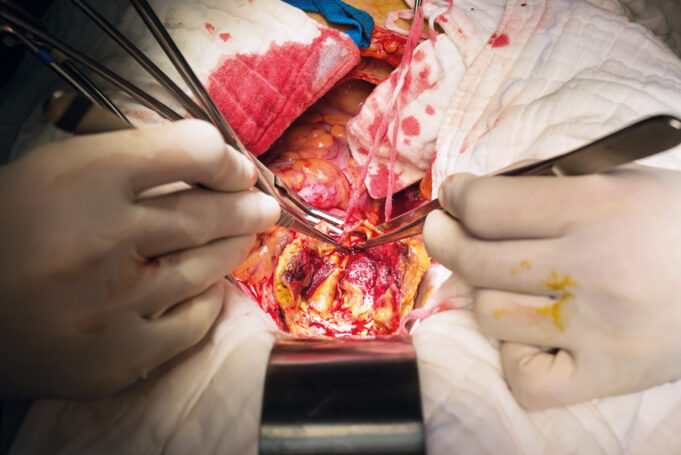
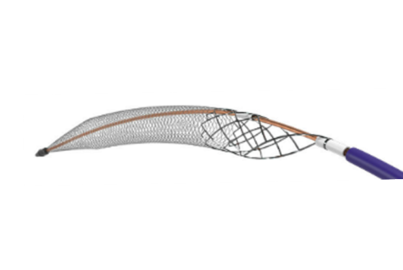
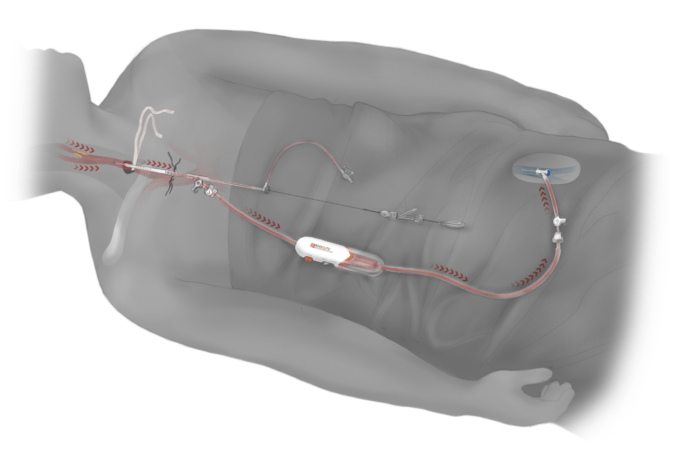
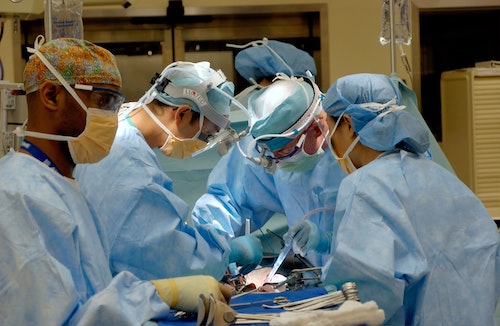
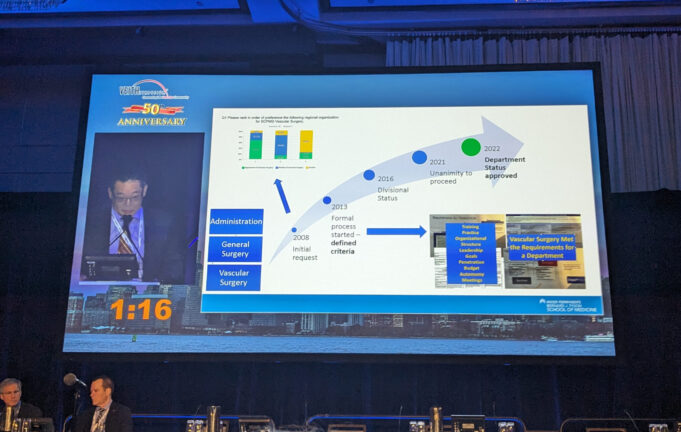
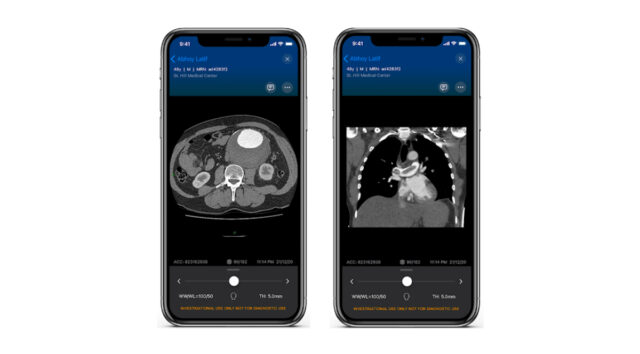
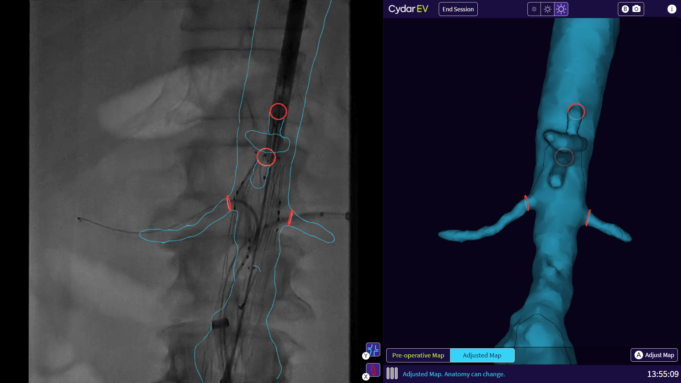
Charles Bailey, MD, George Adams, MD and Vanessa Adams, BS, share their experience with Shockwave Javelin, the first-of-its-kind forward intravascular lithotripsy (IVL) technology that...
In this issue:
Is endovascular dominance undermining surgical training, or are trainees adapting?
Reducing unwanted care through better communication
Meet the 2026 SVS election...
I can feel Brian’s presence hovering over me before he speaks and I know what’s coming. He’s fielded a call from a dialysis patient...
Smoking cessation rates among patients with peripheral artery disease (PAD) undergoing lower extremity revascularization (LER) remain low and largely unchanged, according to a new...
Industry‑sponsored courses can facilitate hands-on exposure and expert-led discussion of new technology, additional operative skill practice and networking with practicing vascular surgeons leading workshops....
Black patients are significantly less likely to undergo inferior vena cava (IVC) filter retrieval after placement compared with white patients, a retrospective study using...
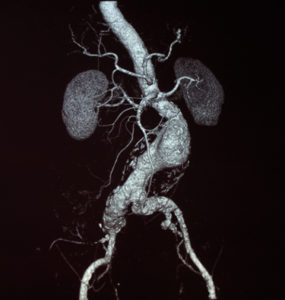
Relying on duplex ultrasound to evaluate suspected left renal vein (LRV) compression may lead to underdiagnosis and delayed treatment of nutcracker syndrome, according to...
A major overhaul of current procedural terminology (CPT) coding for lower extremity revascularization (LER) took effect in January, introducing new codes and allowing physicians to receive dedicated work relative...
Alcohol use disorder (AUD) independently raises the risk of major adverse limb events (MALE) and mortality among patients undergoing lower-extremity revascularization for chronic limb-threatening...
Two Florida Hospital systems — as well as the Cleveland Clinic, Mt Sinai and Johns Hopkins — have been in a contract war with...
Antithrombotic therapy following iliac vein stenting varies widely and long-term outcomes differ significantly depending on the underlying condition, according to new research.
The retrospective study...
In a recent editorial, stakeholders responded to the exclusion of vascular surgical societies from participating in developing updated clinical practice guidelines for the management...
The Society for Vascular Surgery (SVS) has been selected as one of six specialty societies nationwide to receive a competitive grant from the Council...
Female patients had fewer endovascular revascularizations for chronic limb-threatening ischemia (CLTI) compared to male patients but experienced lower risks of mortality and major amputation,...
You pull out the key to open the door to your house. You unlock the door and get inside. Then, several hours later, you...
In this issue:
Biological sex remains a key determinant in CLTI revascularization rates and outcomes
SVS awarded CMSS grant to expand age-friendly vascular ambulatory...
Outpatient vascular care has moved from an alternative site of service to the default setting for much of modern practice. Vascular Specialist marked this...
A systemic review and meta-analysis found transcatheter arterialization of deep veins (TADV) produces higher rates of limb salvage, amputation-free survival (AFS), and wound healing...
The Society for Vascular Surgery (SVS) has published new clinical practice guidelines on the management of blunt thoracic aortic injury (BTAI), reflecting more than...
Becoming an experienced vascular surgeon requires years of rigorous training, profound sacrifice, and sustained dedication. Many accumulate substantial financial debt in pursuit of their...
Carotid endarterectomy (CEA) and transcarotid artery revascularization (TCAR) showed similar perioperative safety and long-term effectiveness in patients with severe cardiac comorbidities, according to a...
A focused education session significantly improved primary care residents’ confidence in diagnosing and managing peripheral artery disease (PAD), a new study found. The findings...
In this issue:
Data support medical therapy first for patients with claudication
SVS Foundation travel grant aims to support early-career female surgeons
Single education...
Women make up only 16% of the vascular surgery workforce, yet female vascular surgeons are 2.05 times more likely to leave practice than their...
The mantra ‘location, location, location’ in property purchases was supposedly emphasized by Lord Harold Samuel, a British real estate tycoon. Location is also important...
Early peripheral vascular intervention (PVI) for claudication is associated with higher rates of progression to chronic limb-threatening ischemia (CLTI) and major adverse limb events (MALE)...
The Centers for Medicare & Medicaid Services (CMS) recently finalized a new Merit-based Incentive Payment (MIPS) System Value Pathway (MVP) specifically for vascular surgery...
The novel I, Claudius, is a historical fiction written by Robert Graves in 1934. Daily life amidst Rome’s political decay during the despotic regimes...
For decades, vascular surgery had few female surgeons. The specialty carved out its territory with increasingly sophisticated techniques, but the operating rooms where surgeons...
The Society for Vascular Surgery (SVS) Foundation has awarded its Vascular Care for the Underserved (VC4U) Project Grant to a study examining the link...
A cohort study using national Medicare data found patients with Alzheimer’s disease and related dementias face significantly higher risks across different outcomes after elective...
Medical editor Malachi Sheahan III, MD, reflects on the release of the CREST-2 results and considers what they may mean for clinical practice.
As you...
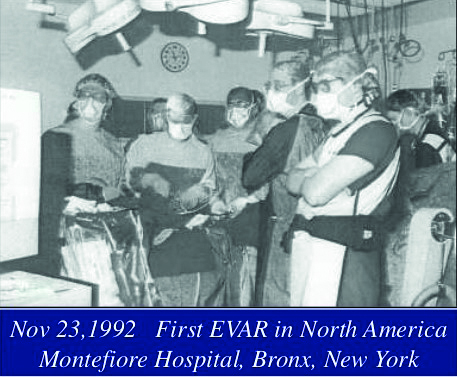
Thomas J. Fogarty, MD, a dedicated surgeon and trailblazing innovator who revolutionized medical technology when he created the first ever minimally invasive surgical device,...
The Association of Program Directors in Vascular Surgery (APDVS), in collaboration with the Society for Vascular Surgery (SVS) Women’s Section and the SVS Foundation,...
A recent study suggests vascular surgeons may be overlooking a crucial element of post-procedural care: blood pressure management. The data, presented at the 2026...
In this issue:
Female surgeons still have lower case volumes when compared to men
VQI-VISION analysis shows premature PAD patients have higher amputation risk
...
Patients with premature peripheral arterial disease (PAD) face a significantly increased risk of mid- and late-term major amputation, according to a new analysis of...
The Society for Vascular Surgery (SVS) has revamped the patient flyers on YourVascularHealth.org—the Society’s patient-facing online patient resource—in an effort to lower the reading level...
Black women with claudication had the highest rate of progression to chronic limb-threatening ischemia (CLTI) after lower extremity revascularization at 180 days, new data...
Outgoing Western Vascular Society (WVS) President Ahmed Abou-Zamzam Jr., MD, provided a possible prescription for value in vascular surgery that goes beyond the monetary...
It's one of the enduring reasons given cross-generationally in answer to the question, “Why vascular?” It goes something along the lines of, “I value...
Shape Memory Medical announced that its Impede embolization plug product family has received certification as a Class III device under the European Union (EU)...
The deadline for applications to the 2026 Society for Clinical Vascular Surgery (SCVS) Young Vascular Surgeons Program has been further extended to Jan. 4...
The Society for Vascular Surgery (SVS) Foundation is marking a milestone in this year's Celebration of Science year-end giving campaign, as former awardees have...
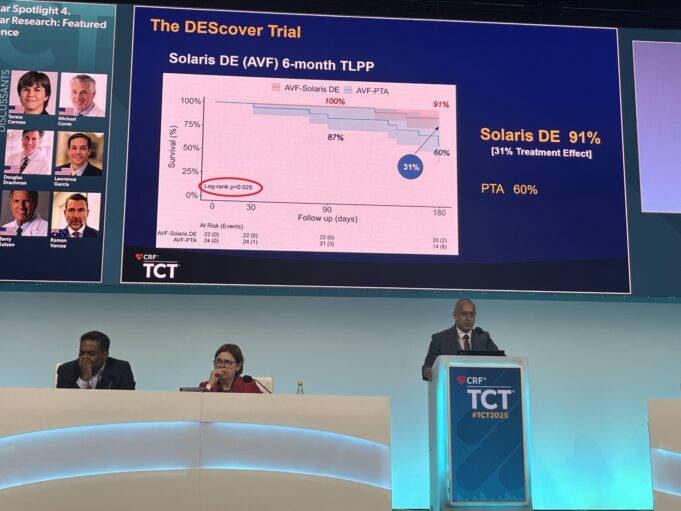
The interim results of a first-in-human trial of a sirolimus-eluting covered stent graft—Solaris DE (Solaris Endovascular)—for the treatment of dialysis access dysfunction demonstrate the...
The Society for Vascular Surgery (SVS) invites authors to submit abstracts for the 2026 Vascular Annual Meeting (VAM 2026) by Jan. 7, 2026, at...
Malachi Sheahan III, MD, has been at the helm of Vascular Specialist for more than seven years. In that time, it is fair to...
A team of leading vascular surgeons in the care of patients with PAD weigh up evidence for the treatment of claudication, calling for research...
In a multi-institutional cohort of patients with intermittent claudication (IC), care deemed inappropriate, or where the risk outweighs the benefit, by the SVS appropriate...
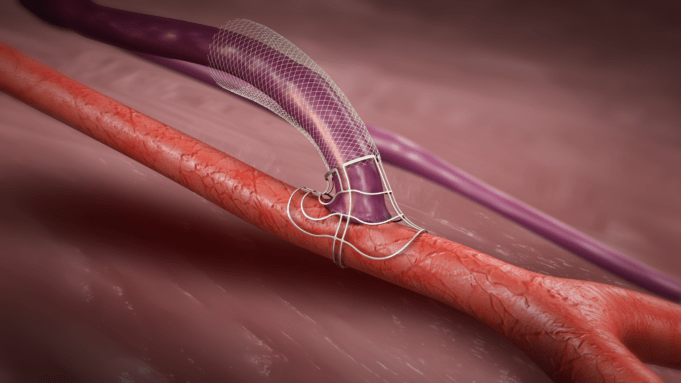
The increasing sophistication of the device space for the treatment of pulmonary embolism (PE) aligned with advances in the deployment of pulmonary embolism response...
In this issue:
CREST-2: Landmark trial finds benefit from carotid stenting
One-year results from IVC stent clinical trial show positive safety and efficacy data
...
Medical editor Malachi Sheahan III, MD, makes the case for surgeon call pay, a remuneration he says should be understood more as compensation than...
The early-bird deadline for registration at the 2026 Southern Association for Vascular Surgery (SAVS) annual meeting in Paradise Island, the Bahamas (Jan. 21–24) falls...
The application deadline for the Society for Clinical Vascular Surgery (SCVS) 2026 Young Vascular Surgeons Program has been extended until midnight Eastern Time on...
Medtronic has announced the first commercial use of its Liberant thrombectomy system, which is indicated for the removal of fresh, soft emboli or thrombi...
Philips has announced the expanded commercial availability of LumiGuide 3D device guidance—the much-heralded first real-time artificial intelligence (AI)-enabled light-based navigation solution for image-guided therapy—across...
The National Institutes of Health (NIH) has approved funding for the WARRIORS international randomized trial comparing endovascular aneurysm repair (EVAR) with watchful waiting in...
Vascular specialists have been intensifying efforts to combat tobacco use through the Society for Vascular Surgery Patient Safety Organization (SVS PSO), which is leading...
Drug-coated balloon (DCB) angioplasty appears to offer similar benefits as plain old balloon angioplasty (POBA), with or without stenting, for patients with chronic limb-threatening...
Chinmaya Shelgikar, MD, discusses the benefits of vascular surgeons performing geniculate artery embolization.
Geniculate artery embolization (GAE) is an intra-arterial, catheter-based procedure used to treat chronic...
Just a few years ago, it was commonly accepted that training in complex endovascular aortic operations (e.g. physician-modified endografts , use of in-situ laser...
The sixth cohort of the Society for Vascular Surgery (SVS) Leadership Development Program (LDP) gathered Oct. 19–20 in Rosemont, Illinois, for a capstone workshop...
“We are the first major U.S.-based surgical society to present this topic to our membership. Once again, vascular surgery is leading the way,” said...
This advertorial is sponsored by Stryker/Inari Medical.
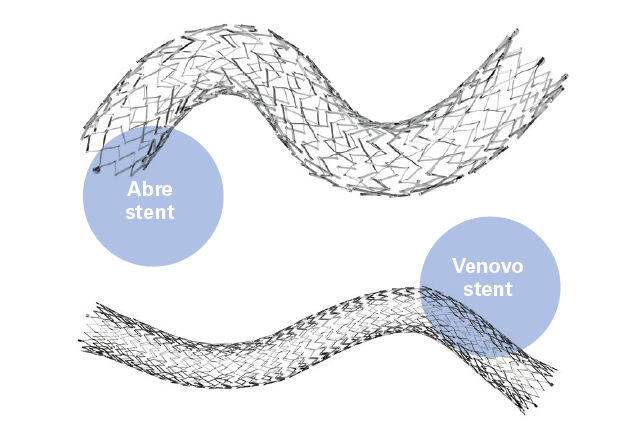
As venous stenting becomes more common, the focus must now shift to what occurs after the procedure, according...
Clyde F. Barker, MD, a towering figure in American surgery and a transformative leader at Penn Medicine, died peacefully at his home on Oct....
Froedtert Hospital has become the first hospital in Wisconsin to receive national verification as a Comprehensive Inpatient Vascular Center through the American College of...
The Society for Vascular Surgery (SVS) Foundation’s 2025 annual report details a year of significant progress in research, education and advocacy aimed at improving...
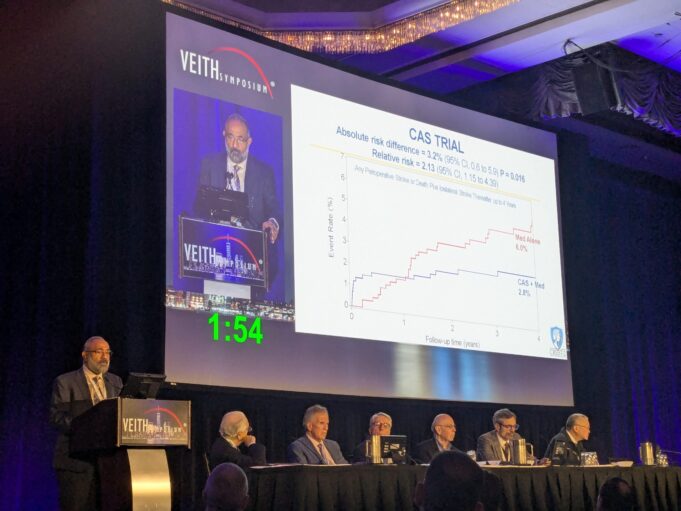
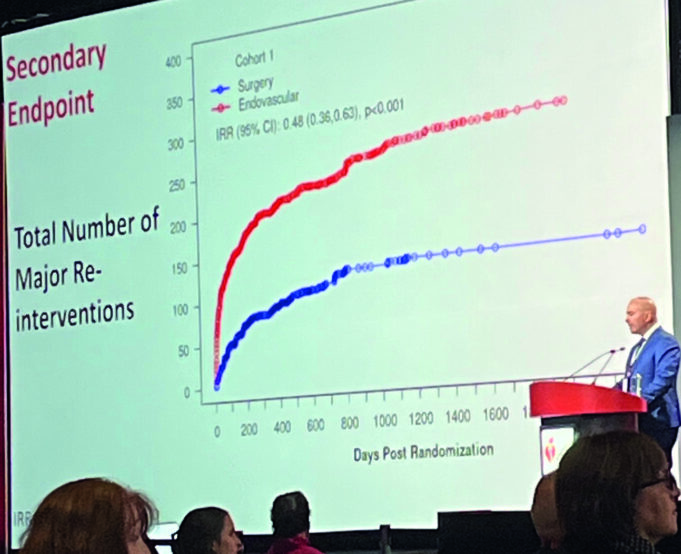
The National Institutes of Health (NIH)-funded CREST-2 study has found that, for people with high-grade asymptomatic carotid artery stenosis who have not experienced recent...
The international CREST-2 study has found that, for people with severe carotid artery narrowing who have not experienced recent stroke symptoms, a minimally invasive...
A systematic review and meta-analysis of more than 300 original investigations on atherectomy in peripheral arterial disease (PAD) highlights “overall favorable” clinical outcomes. This...
This advertorial is sponsored by Abbott.
“We now have something on label and FDA approved that we can use as a first-line therapy for...
Terumo Aortic today announced enrollment of the first patient in the Fenestrated Treo pivotal investigational device exemption (IDE) study in the U.S. This study...
A focused session at the 2025 Vascular Interventional Advances (VIVA) conference (2–5 November, Las Vegas, USA) homed in on the latest findings from the...
RapidAI today announced the US Food and Drug Administration (FDA) clearance of Aortic Management, part of Rapid Aortic, which the company describes as "a...
enVVeno Medical has announced that it received an “unfavorable” decision from the Food & Drug Administration (FDA) in response to its supervisory appeal of...
enVVeno Medical has announced that it has received an "unfavorable" decision from the Food & Drug Administration (FDA) in response to its supervisory appeal...
This advertorial is sponsored by Shockwave Medical.
Three vascular surgeons discuss how and when they deploy the transformative Shockwave Javelin first-of-its-kind Forward Intravascular Lithotripsy (IVL)...
ViTAA Medical today announced that the Food and Drug Administration (FDA) has granted 510(k) clearance for AiORTA Plan, which the company describes as a...
Elucid has announced the launch of its PlaqueIQ image analysis software for the quantification and classification of plaque morphology in the carotid arteries. According to...
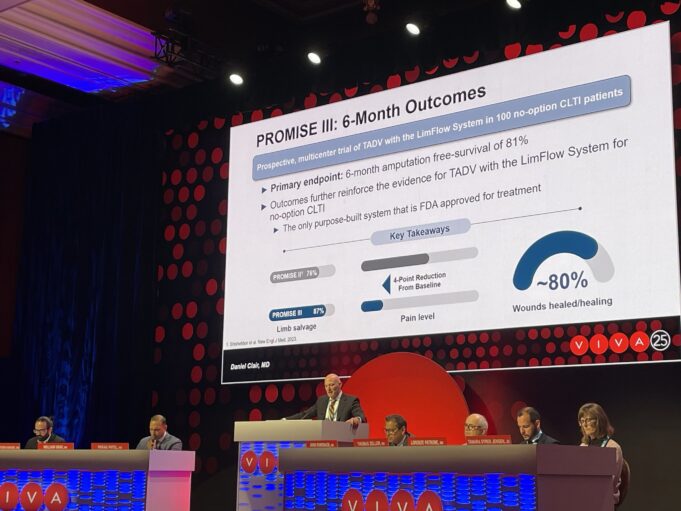
Six-month outcomes among no-option chronic limb-threatening ischemia (CLTI) patients treated using transcatheter arterialization of the deep veins with the LimFlow system (Stryker/Inari Medical) in...
Thirty-day results from the PERFORMANCE III study of integrated embolic protection (IEP) in transcarotid artery revascularization (TCAR) demonstrate “very low” 30-day event rates. Sean...
One-year outcomes in the ROADSTER 3 post-approval study to evaluate the safety and effectiveness of transcarotid artery revascularization (TCAR) using the Enroute stent and...
Stay updated on the latest advancements in wound care with the Society for Vascular Surgery (SVS), Society for Vascular Nursing (SVN), and American Podiatric...
Twelve-month results from the clinical trial assessing a dedicated venous stent for the treatment of symptomatic inferior vena cava (IVC) obstruction with or without...
InterVene recently announced it has received 510(k) clearance from the US Food and Drug Administration (FDA) for the Recana thrombectomy catheter system, which is...
Twelve-month results from the clinical trial assessing a dedicated venous stent for the treatment of symptomatic inferior vena cava (IVC) obstruction with or without...
Vein mapping for lower extremity bypass in patients with chronic limb-threatening ischemia (CLTI) remains unchanged two years after the BEST-CLI, "revealing the difficulty in...
Former Society for Vascular Surgery (SVS) President Anton N. Sidawy, MD, FACS, DFSVS, became the 106th president of the American College of Surgeons (ACS)...
Proposals for the new "Hot Topics” session at the 2026 Vascular Annual Meeting (VAM) recently drew in a slew of responses for the inaugural...
In 1979, after three years of group practice, I opened an office as a solo vascular surgeon. With a wife, three children and a...
During his own recent presidential address on home turf, Patrick Muck, MD, invoked immortable words from the inaugural address of President John F. Kennedy...
“How did an ineffective and costly intervention become routine care in the NHS ?” Ian Chetter, MBChB, chair of surgery at Hull York Medical...
Prespecified interim analysis data from the first 50 patients in RAPID-PE have demonstrated "excellent safety" and "remarkably efficient lab times" in patients receiving on-the-table...
Matthew Corriere, MD, believes a major shift is needed in how clinicians communicate and make decisions with their patients. During the 2025 distinguished address...
A novel physician-modified endograft (PMEG) technique for the treatment of thoracoabdominal and complex abdominal aortic aneurysms (TAAA and AAA) involving delayed-release diameter reduction to...
New findings chronicling progress with both the Gore Excluder thoracoabdominal branch endoprosthesis (TAMBE)— approved by the Food and Drug Administration (FDA) last year—and Gore...
A readability analysis of 15 publicly available patient education materials from the SVS found that all but three exceeded the 8th-grade reading level, the...
A retrospective analysis of 64 patients treated between 2022 and 2025 using a novel mechanical thrombectomy system to treat dialysis access thrombus showed function...
The use of mechanical thrombectomy, specifically computer-assisted vacuum thrombectomy (CAVT) using the 16Fr Lightning Flash system (Penumbra), with anticoagulation achieves superior reduction in right...
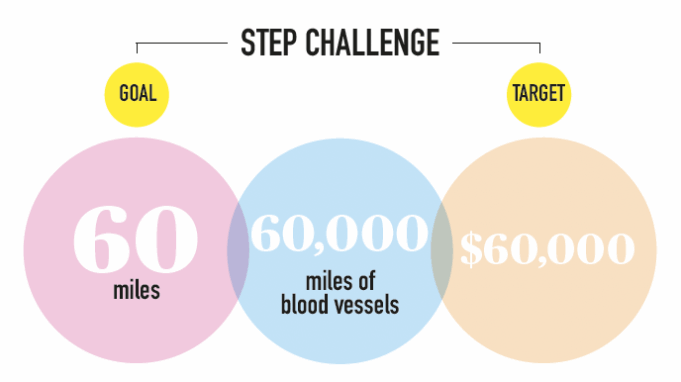
The Stanford Division of Vascular Surgery launched its first in-person SVS Foundation Vascular Health Step Challenge on Sept. 7, bringing together more than 100...
The Society for Vascular Surgery (SVS) Foundation will formally establish the Enrico Ascher Vascular Innovation Institute as a new entity under its research and...
In 1954, Dr. Michael DeBakey transformed vascular surgery with a homemade Dacron graft sewn on his wife’s sewing machine. His spirit of innovation laid...
UMass chief peers down the lens of an if-not radiation-free complex aortic procedural future, then one in which the methods of today will look...
In this issue:
TAMBE and TBE: Evolving experiences and piecing together channels of inquiry
Randomized data debunks widely used negative pressure wound therapy
The...
This advertorial is sponsored by Stryker/Inari Medical.
How a finely tuned stent surveillance program plays a crucial role in raising the alarm of worsening symptoms...
WFVS secretary general details how the organization has evolved in recent years, seeing gains in representation and access to education among members from underserved...
Shape Memory Medical has reached 50 per cent enrolment in its ongoing AAA-SHAPE pivotal trial, the company announced last week.
AAA-SHAPE is a prospective,...
Results from a multicenter randomized controlled trial (RCT) indicate that pre-emptive inferior mesenteric artery (IMA) embolisation during endovascular aneurysm repair (EVAR) does not significantly...
Medtronic recently announced it has received Food and Drug Administration (FDA) labelling approval for the Endurant stent graft system, by adding ruptured abdominal aortic...
Data from the AMDS PERSEVERE and PROTECT trials—both assessing the use of the Ascyrus medical dissection stent (AMDS, Artivion)—were presented in late-breaking science presentations...
Nurea's PRAEVAorta 2 artificial intelligence (AI)-powered software has received US Food and Drug Administration (FDA) 510(k) clearance, a company press release reveals.
According to Nurea,...
Researchers have shown that quiescent-inflow single-shot (QISS) magnetic resonance imaging (MRI) is able to identify more below-the-knee vessel segments than digital subtraction angiography (DSA)...
Malachi Sheahan III, MD, chair of the Department of Surgery and chief of vascular surgery at Louisiana State University in New Orleans, is the...
Medtronic has announced the full distribution of the Neuroguard integrated embolic protection (IEP) system for carotid artery disease following its limited market distribution, the...
Alik Farber, MD, surgeon-in-chief at Boston Medical Center and James Utley professor and chair of surgery at Boston University Chobanian and Avedisian School of...
Terumo Aortic and Bentley today announced their partnership in a clinical study in the USA. The objective is to obtain US Food and Drug...
The Society for Vascular Surgery (SVS) held its inaugural Advocacy and Leadership Conference Sept. 14–16, drawing vascular surgeons, trainees and healthcare advocates from across...
Transcatheter arterialization of deep veins (TADV) done without the coiling of venous outflow branches achieved sufficient distal perfusion after three months among “no-option” chronic...
The Society for Vascular Surgery (SVS) continues to expand its global footprint through its 13 international chapters, fostering collaboration and visibility for vascular surgeons...
The Society for Vascular Surgery (SVS) will take part in an upcoming webinar titled “Breaking the Habit, Saving Lives: The National Quality Initiative Push...
As vascular surgeons, we strive to practice comprehensive vascular surgery care. This entails the medical and surgical management of patients with a wide range...
In these challenging days of surviving private vascular practice, I’m sure we’re all considering our options. With rising overheads and reimbursement cuts, considering a...
Venita Chandra, MD, clinical professor of surgery at Stanford Health Care in Stanford, California, will serve as a co-principal investigator for the upcoming investigational...
Teams formally defined as specializing in chronic limb-threatening ischemia (CLTI) were associated with a decreased risk of major amputation among a subset of BEST-CLI...
Health Canada has authorised the Esprit BTK everolimus-eluting resorbable scaffold system for the treatment of below-the-knee (BTK) chronic limb-threatening ischaemia (CLTI), Abbott revealed today...
Samantha Minc, MD, MPH, associate professor of surgery at Duke University, has been awarded a National Institutes of Health (NIH) R03 grant to support...
Vascular Specialist readers may wonder why I am now raising this topic amid current challenges facing vascular surgery, like governmental concerns, low reimbursement, specialty...
Ross Milner, MD, the Louis Block professor of surgery and chief of vascular surgery at the University of Chicago, became the new president of...
Researchers have reported comparable five-year outcomes of the Gore Excluder iliac branch endoprosthesis (IBE; Gore) in both the investigational device exemption (IDE) and GREAT...
David Rigberg, MD, clinical professor of surgery, at the UCLA Gonda School of Medicine in Los Angeles, has taken over as president of the...
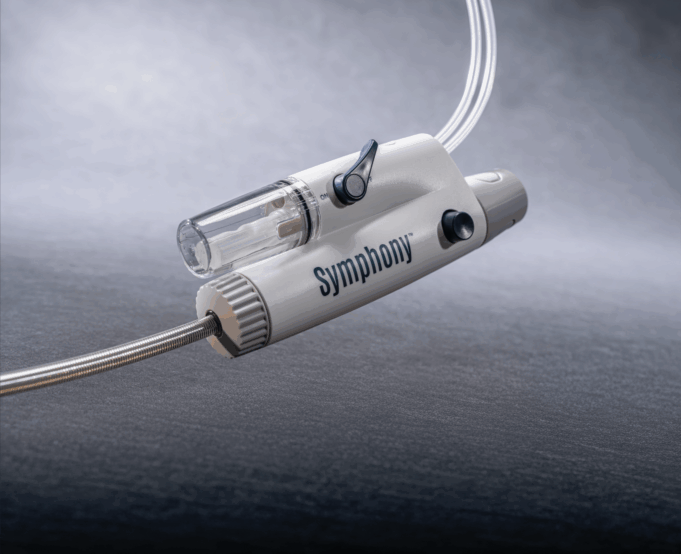
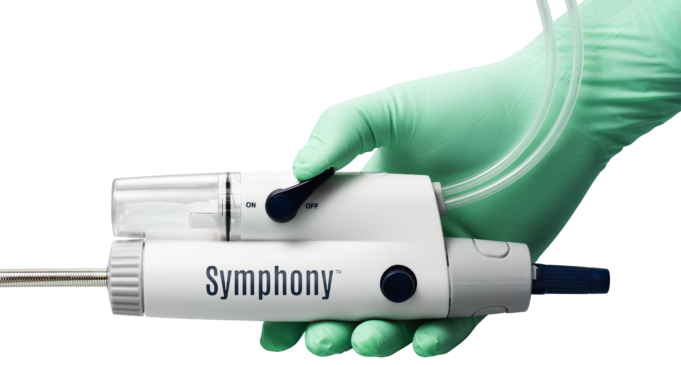
Imperative Care has announced efficacy and safety results from the pivotal SYMPHONY-PE trial evaluating the company’s Symphony thrombectomy system in the treatment of acute...
Terumo Aortic today announced what it describes as a "significant policy milestone" with the Centers for Medicare & Medicaid Services (CMS) establishing a new...
Venous valve developer enVVeno Medical has announced that it will file a request for supervisory appeal of the "not-approvable" letter from the Center for...
This is the latest monthly update on a few of the critical issues facing the SVS.
Changing SVS leadership demographics
Over the last several years,...
The Society for Vascular Surgery (SVS) recently published comprehensive update to its clinical practice guidelines (CPGs) for the management of intermittent claudication (IC), urging...
The Society for Vascular Surgery (SVS) is ramping up its national Highway to Health campaign to educate the public about peripheral arterial disease (PAD)...
In this issue:
Enduring PROMISE: Real-world outcomes study suggests results continue to flow from transcatheter arterialization of the deep veins
SVS Highway to...
Gregorio A. Sicard, MD, a transformative figure in vascular surgery and a revered mentor to generations of surgeons, has died. He was 81.
Sicard was...
First retrospective analysis shows comparable outcomes data to those found in PROMISE II pivotal trial, authors report.
The first post-approval multicenter analysis of so-called “no-option”...
It “takes village” to build a community like the one that the Eastern Vascular Society (EVS) has grown into, outgoing president Brajesh K. Lal,...
Microbot Medical today announced that the Food and Drug Administration (FDA) has granted 510(k) clearance for the Liberty system, the first FDA cleared single...
Shape Memory Medical today announced the completion of patient enrollment in the EMBO postmarket surveillance (EMBO-PMS) registry, the company’s prospective, multicenter registry of the...
Benjamin Jackson, MD, chief in the Division of Vascular and Endovascular Surgery at Lehigh Valley Heart and Vascular Institute in Allentown, Pennsylvania, became the...
The SVS Complex Peripheral Vascular Interventions (CPVI) Skills Course has become a fixture on the vascular surgery calendar over the past couple of years....
The Society for Vascular Surgery (SVS) Foundation reminds eligible applicants of the upcoming submission deadline for the Vascular Care for the Underserved (VC4U) project...
The PAD Pulse Alliance—a coalition of leading medical societies that includes the Society for Vascular Surgery (SVS) dedicated to improving vascular health—is urging patients,...
Imperative Care has today announced Food and Drug Administration (FDA) 510(k) clearance of its Symphony thrombectomy system to treat pulmonary embolism (PE).
This clearance expands...
The Food and Drug Administration (FDA) recently announced that it has begun daily publication of adverse event data from the FDA Adverse Event Reporting...
Drug-coated balloons and stents were not associated with reduced risk of amputation or improved quality of life compared with uncoated devices in the SWEDEPAD...
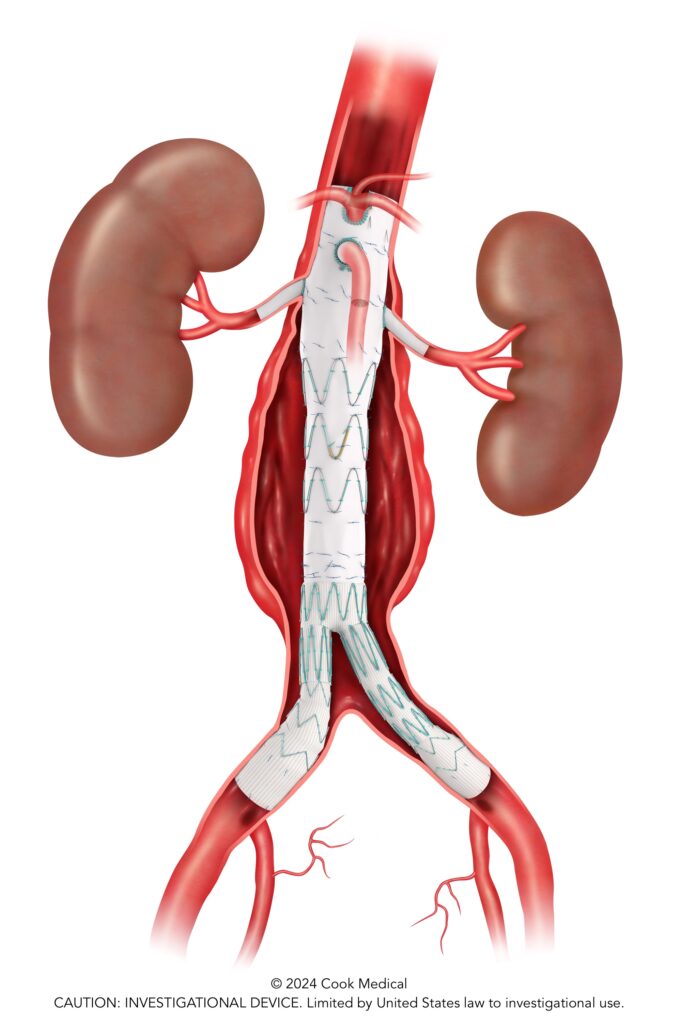
Cook Medical has enrolled the final patient in the global clinical study of its Zenith Fenestrated+ endovascular graft (ZFEN+), signifying completion of patient recruitment in...
The Journal of Surgery (JVS) has appointed Audra Duncan, MD, of Western University in London, Ontario, Canada, as its new editor-in-chief.
Duncan will lead the...
Boston Scientific has recalled its Carotid Wallstent Monorail endoprosthesis owing to a “manufacturing defect” that has led to devices having an inner lumen that...
A recent French study suggests smoking cessation in patients with peripheral arterial disease (PAD) is achievable with intensive and appropriate management, with the prescription...
Congress entered summer recess after passing the One Big Beautiful Bill Act (OBBBA), which was signed into law by President Donald J. Trump on...
The speed with which artificial intelligence (AI) is beginning to envelope aspects of the human experience strikes many as jarring. Its entrance into the...
Abbott today announced it has received CE mark approval in Europe for the Esprit BTK everolimus-eluting resorbable scaffold system (Esprit BTK system), a technology...
The vascular surgery field received a rare moment in the public spotlight recently as President Donald J. Trump was diagnosed with chronic venous insufficiency...
The Food and Drug Administration (FDA) has granted approval for Cook Medical to initiate an investigational device exemption (IDE) study on the Advance Evero...
The Food and Drug Administration (FDA) has issued a letter to Envveno Medical stating that its VenoValve technology is "not-approvable," a company press release...
Vikram S. Kashyap, MD, pushes back against a recent editorial from Vascular Specialist medical editor Malachi Sheahan III, MD, in which the latter called...
Whether you're a seasoned vascular surgeon or a first-time attendee, Society for Vascular Surgery (SVS) Program Committee Chair Jason Lee, MD, and Postgraduate Education...
Comparative analysis looks at stroke-related disability and mortality after three modes of carotid revascularization.
Strokes after transfemoral carotid artery stenting (TFCAS) were “the most disabling...
The Society for Vascular Surgery (SVS) Foundation is placing a spotlight on its “Legacy Teams”—those who have gone the extra mile, literally!—to make the...
The THRIVE study - which shows that computer-assisted vacuum thrombectomy (CAVT™) is associated with reduced related readmissions and complication rates in the management of...
Healthcare professionals from multiple disciplines are embracing the Wound Care Curriculum, a flexible online learning experience tailored to meet the evolving needs of vascular...
You don't have to be too far down the path to get started. That’s the message William Shutze, MD, chair of the Society for...
During the upcoming year, I plan to provide a monthly summary of critical issues the SVS is facing to keep you better informed. There...
The Food and Drug Administration (FDA) has approved the expansion of Aquedeon Medical's investigational device exemption (IDE) clinical trial evaluating the Duett vascular graft system,...
News that U.S. President Donald J. Trump has a vascular condition may have simultaneously revealed that a substantial seven-figure vascular surgery branding campaign was...
In this issue:
Artificial intelligence: Where it lands today and how it might impact the vascular future
In wake of President Trump’s chronic venous...
The glut of early fall regional vascular society annual meetings is just around the corner, with no fewer than five slated for the month...
After two years of careful deliberation, the Society for Vascular Surgery (SVS) Executive Board (EB) has voted against pursuing the creation of a free-standing...
A post-hoc comparison of the BEST-CLI and PREVENT III multicenter, prospective, randomized controlled trials (RCTs) aimed at evaluating outcomes of vein bypass in chronic...
The early bird pricing deadline for this year’s SVS Coding & Reimbursement Workshop is tomorrow.
With significant coding changes going into effect for vascular surgery...
Ten-year data highlight the long-term efficacy and durability of the Endurant stent graft (Medtronic) in abdominal aortic aneurysm (AAA) patients who survived beyond five...
There's no place like home within the newly established SVS Advanced Practice Provider (APP) Section, where vascular advanced practice registered nurses (APRNs) and physician...
The Society for Vascular Surgery (SVS) Foundation has extended the deadline for a Vascular Care for the Underserved (VC4U) project grant focused on women's...
Inari Medical—now part of Stryker—has announced the launch of its next-generation InThrill thrombectomy system.
The company stated in a recent press release that this is...
The Society for Vascular Surgery (SVS) is gearing up for its inaugural Advocacy and Leadership Conference, taking place Sept. 14–16, in Washington, D.C. With...
AngioDynamics today announced enrollment of the first patient in AMBITION BTK, a randomized study of the Auryon atherectomy system in patients with below-the-knee chronic...
An anonymous vascular resident offers some advice for dealing with a troubling case in the latest edition of our Corner Stitch column by and...
Get ready to join the Society for Vascular Surgery (SVS) Foundation this September for the 2025 Vascular Health Step Challenge.
Hosted during National Vascular Disease...
Registration is now open for the Society for Vascular Surgery (SVS) Complex Peripheral Vascular Interventions (CPVI) Skills Course, taking place from Sept. 20–21 in...
InterVene announced today that the first patient has been treated with its Recana thrombectomy catheter system for venous in-stent restenosis (ISR).
The company shares...
The Society for Vascular Surgery has highlighted findings from a comprehensive 20-year clinical study that demonstrate treatment of functional popliteal artery entrapment syndrome—particularly in...
The Medicines and Healthcare products Regulatory Agency (MHRA) has today announced important new steps to secure access for patients to the latest medical technologies...
The Society for Vascular Surgery (SVS) Foundation is currently accepting applications for its 2025 Vascular Care for the Underserved (VC4U) project grants until Aug....
When is the right time to start a family in vascular surgery? That was a core question posed during the 2025 Vascular Annual Meeting...
“How many programs conducted in-person interviews this year?” “Which programs conducted in-person interviews?” “Oh wow, I didn’t realize that many programs were conducting in-person...
Registration for the annual Society for Vascular Surgery (SVS) Coding and Reimbursement Workshop—and optional evaluation and management (E/M) coding workshop—opened July 9 with early-bird...
With another Vascular Annual Meeting (VAM) in the books, that means the flag unfurls on a new presidential year. And that means Philadelphia-based vascular...
The Society for Vascular Surgery (SVS) has announced a new partnership with group purchasing organization (GPO) Provista in an effort to help provide...
Getinge recently announced that it has received Food and Drug Administration (FDA) premarket approval (PMA) for the iCast covered stent system. The device is...
A five-year prospective cohort study—using longitudinal registry data from two population-based screening trials—has found that high-dose statin use was associated with decreased abdominal aortic...
Medtronic today announced that the first patient has been enrolled in the Peripheral Onyx liquid embolic (PELE) clinical trial, which will evaluate the safety...
In this issue:
Vascular unity: ‘Let’s stop standing on the sidelines while others define what vascular care looks like’
Vascular surgery history: Putting...
Vascular surgeons should come together to present a unified voice in the world of medicine—it’s time for a change, to recommit, build bridges and...
Medtronic today announced it has entered into an exclusive U.S. distribution agreement with Japan-based Future Medical Design Co (FMD) to sell specialty and workhorse...
Cook Medical has announced that its Zenith iliac branch device (ZBIS) is now commercially available in the U.S. with Food and Drug Administration (FDA)...
Dear editor,
As members of the Society for Vascular Nursing (SVN) Board of Directors, the Society for Vascular Surgery (SVS) Advanced Practice Provider (APP) Section,...
The people, discoveries, innovations and events that brought the vascular surgery profession to where it is today formed the bedrock of the subject matter...
“Spinal fluid drainage be considered when the risk of spinal cord injury from the procedure is greater than the risk of severe complications from...
The Society for Vascular Surgery (SVS) Patient Safety Organization (SVS PSO) will host a webinar on Tuesday, July 15, at 1 p.m. Eastern Time...
Humacyte announced today that Symvess has been awarded Electronic Catalog (ECAT) listing approval from the U.S. Defense Logistics Agency.
ECAT is an internet system...
Gore today announced CE-mark approval of an expanded indication for the Gore Viabahn VBX balloon-expandable endoprosthesis (VBX stent graft) when used as a bridging...
“We want to change global standards. What we’re doing isn’t working. Patients are getting hurt and devices are failing—we want to be able to...
Vascular surgeons must present a unified front in order to address structural and financial challenges facing the specialty, Society for Vascular Surgery (SVS) President-Elect...
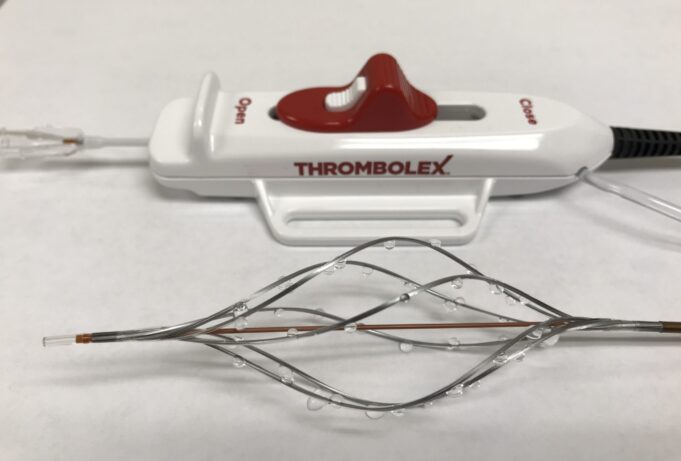
Thrombolex recently announced the publication of RESCUE-II study results in JACC: Advances.
The RESCUE-II study was a single-centre, prospective study evaluating the safety and...
Cook Medical has initiated a Class I recall of its Beacon Tip 5F angiographic catheter.
This follows reports of tip separation that could result...
The Society for Cardiovascular Angiography & Interventions (SCAI) has released new, evidence-based clinical practice guidelines to support the treatment of chronic venous disease (CVD).
Published...
StentIt has announced the successful implantation of its Resorbable Fibrillated Scaffold (RFS). As part of the VITAL-IT 1 study, patients with chronic limb-threatening ischemia...
Vivasure Medical has announced the submission of a premarket approval application to the Food and Drug Administration (FDA) for its PerQseal Elite vascular closure...
The final day of the 2025 Vascular Annual Meeting (VAM) in New Orleans honored three winners in the championship round of the SVS Poster...
Results from the CLN-PRO-V007 pivotal phase 3 clinical trial of the acellular tissue engineered vessel (ATEV; Humacyte) in arteriovenous access for patients at high...
Attendees of the 2025 Vascular Annual Meeting (VAM), Vascular Quality Initiative (VQI) Annual Meeting, and Society of Vascular Nursing (SVN) Annual Conference are reminded...
The Society for Vascular Surgery (SVS) invites members to register for its inaugural Advocacy and Leadership Conference, taking place Sept. 14–16, in Washington, D.C....
“We were really excited to know that the SVS is supporting our research,” Sujin Lee, MD, told Vascular Specialist on receiving the SVS Foundation...
As vascular surgeons advance in their careers, many begin to consider alternative professional paths that draw upon their clinical expertise, while offering greater flexibility,...
This advertorial is sponsored by Laminate Medical.
Arteriovenous fistula (AVF) creation remains one of the most frequent, yet frustrating procedures performed by vascular surgeons. While...
Society for Vascular Surgery (SVS) Vice President Linda Harris, MD, extolled the values of “co-elevation” in her presidential address at the 7th annual Women’s...
InspireMD has been granted premarket approval (PMA) from the Food and Drug Administration (FDA) for its CGuard Prime carotid stent system in the U.S.—news...
Gore has announced that the Gore Tag thoracic branch endoprosthesis (TBE) is now approved by the Food and Drug Administration (FDA) for use in...
Michele Piazza, MD, informed attendees of the 2025 Vascular Annual Meeting (VAM) that increased experience with the E-nside (Artivion) off-the-shelf preloaded inner branch endograft for...
Penumbra has announced the completion of enrollment in the STORM-PE clinical trial.
The pivotal, prospective, multicenter randomized controlled trial enrolled 100 patients to evaluate computer...
Inquis Medical has announced that its Aventus thrombectomy system has received 510(k) clearance from the US Food and Drug Administration (FDA) for an expanded...
“The reason to avoid a heart and vascular paradigm is there really is no paradigm. There's no set standard for how these things...
“Go back home and get a vascular center or get a vascular department, or, you know, assess the compensation results and, at...
Recent THRIVE study data show that Penumbra’s computer-assisted vacuum thrombectomy (CAVT) technology not only has the potential to improve outcomes for lower extremity acute...
The Society for Vascular Surgery (SVS) has elected Andres Schanzer, MD, as the incoming vice president and Malachi G. Sheahan III, MD, as the...
Implantation of the VenoValve (enVVeno Medical) continues to promote stabilization of symptoms in patients with deep venous reflux at two-year follow-up. This is the...
The people, discoveries, innovations and events that brought the vascular surgery profession to where it is today will form the subject matter for this...
A study aimed at assessing the impact of a pandemic-related switch to virtual recruitment from in-person interviews on the Match for vascular surgery residents...
There is an urgent need for epidemiological studies to map out the prevalence of chronic limb-threatening ischemia (CLTI)—and peripheral arterial disease (PAD) in general—in...
In this issue:
Transatlantic multicenter experience highlights effectiveness of total endovascular aortic repair with F/BEVAR but 5% risk of major stroke
SVS president will...
“A lot of solutions to your problems exist in somebody else's operating room, or somebody else's cath lab, but you never actually go there.”...
One of the singular highlights of VAM 2025—the presidential introduction and address—will take place this morning from 11 a.m.–12 p.m. on Morial CC’s First...
Staged total endovascular aortic repair (TEAR) utilizing arch branched and thoracoabdominal fenestrated and branched endografts is effective, but identified predictors of morbidity and mortality—including...
Don't miss the chance to witness the creativity and hard work of the Poster Competition participants as they compete for top honors, taking place...
Results from the CLN-PRO-V007 randomized controlled trial of Humacyte’s acellular tissue engineered vessel (ATEV) are set to be shared today during VAM 2025 Plenary...
Sherene Shalhub, MD, set out a new biologically informed framework that “captures the diverse spectrum of arterial fragility and repair outcomes across inherited and...
An evaluation of trends in the utilization of carotid artery stenting (CAS) versus carotid endarterectomy (CEA) has found there to be “no major change”...
“We're living in a new world order” when it comes to “no-option” chronic limb-threatening ischemia (CLTI) patients, says Anahita Dua, MD. In recent years,...
SVS members in good standing can still cast their votes for key leadership positions and important bylaw changes in the 2025 elections to have...
Today's Plenary Session 4—Hosted in Great Hall A, First Floor from 10:45 a.m.–12 p.m.—will feature a presentation of five-year findings from the VQI-VISION database...
Some of the enduring controversies in the management of asymptomatic severe carotid stenosis will underpin an educational session set to take place today.
The session...
In this issue:
One-year TAMBE data highlight low mortality, importance of renal branch assessment
Transcatheter arterialization of the deep veins: Comparative analysis probes...
Concerns were raised yesterday’s Plenary Session 2 over the recent decline in Medicare reimbursement at a national level. Querying current figures, speakers revealed discrepancies...
The four-branch Gore Excluder thoracoabdominal branch endoprosthesis (TAMBE) device demonstrated low mortality at one year with renal artery occlusion being the predominant device-related event...
The vascular surgery world is used to pivoting on a dime. So it was as VAM 2025 was about to commence yesterday morning,...
In this issue:
Crawford Forum: Empowering vascular surgeons
SVS Foundation Resident Research Award: Targeting highly complex process of calcification
VTE: Evolving landscape of interventions for...
Since the 2018 Accreditation Council for Graduate Medical Education (ACGME) reduction of core surgical requirements from 24 to 18 months during integrated vascular surgery...
Join your colleagues for engaging Women’s Networking Event on Thursday evening at 7 p.m. at the 2025 Vascular Annual Meeting (VAM) in New Orleans...
Today, 2025 Vascular Annual Meeting (VAM) attendees are encouraged to engage in an eye-opening experience at Touchpoint@VAM for an interactive event that will showcase...
Medical students attending the 2025 Vascular Annual Meeting (VAM) will have two unique opportunities to deepen their understanding of vascular surgery through sessions designed specifically...
Results of a first-in-human study of non-invasive, robotic-assisted high intensity focused ultrasound (HIFU) for the treatment of peripheral arterial disease (PAD), to be presented...
A number of topics relating to pulmonary embolism (PE) management—including lytic therapy versus thrombectomy, risk stratification and device selection—will feature this afternoon during an...
Philip P. Goodney, MD, is unveiling a pivotal monitoring tool during the Vascular Quality Initiative (VQI) Annual Meeting, VQI@VAM, today—the Long-Term EVAR Assessment and...
As we come together for VAM 2025, I want to take a moment to reflect on the remarkable progress we’ve made over the past...
This year’s E. Stanley Crawford Critical Issues Forum at the 2025 Vascular Annual Meeting (VAM) in New Orleans, from 11:15 a.m.–12:15 p.m., on the...
How does the cost of treating critical limb-threatening ischemia (CLTI) in the U.S. stack up to cancer care? That was the question at the...
Welcome! Our city is humid, crumbling and vaguely supernatural. Make the right choices and you will never forget your time here. Double that if...
In this issue:
Helping OBL vascular surgeons fight the squeeze
‘Rise together’: Harris urges unity in Women’s Vascular Summit presidential address
Senior surgeons:...
The open surgical approach will prove to be “slightly superior” to endovascular intervention in terms of cost per life year gained, while the latter...
The Vascular Quality Initiative (VQI), in collaboration with the Society for Vascular Surgery (SVS), will convene its ninth annual conference (June 3–4) during this...
It’s a bit like realigning the narrative, ever so slightly. “I want us to feel, as a collective specialty, that pediatric vascular care falls...
The Society for Vascular Surgery (SVS) Vascular Quality Initiative (VQI) is hosting a monthly webinar series throughout 2025 on smoking cessation, which so far...
Jack L. Cronenwett, MD, considered the “Grandfather of the VQI,” will retire as the chief medical officer (CMO) for Fivos, the medical data solutions...
SVS Section on Ambulatory Vascular Care (SAVC) set to announce set of new initiatives designed to help office-based lab (OBL) vascular surgeons combat rising...
"The path from initial trials to meaningful clinical data will not be easy,” writes Fedor Lurie (Toledo, USA) of new venous valve technologies.
There is...
The Society for Vascular Surgery (SVS) has issued a comprehensive update to its clinical practice guidelines for the management of intermittent claudication, urging clinicians...
Terumo Interventional Systems has announced the early commercial availability of its US Food and Drug Administration (FDA)-approved Roadsaver carotid stent system. Indicated for use...
Malachi Sheahan III, MD, produces notes on a quixotic quest to find meaning in one of medicine’s most meaningless endeavors.
Here’s how it happens. One...
The Society for Vascular Surgery (SVS) announced the nominees for the vice president and secretary positions in April. Voting in this year's SVS elections...
The Society for Vascular Surgery (SVS) is entering a transformative period as Vice President Linda Harris, MD, transitions to the role of SVS president-elect,...
A new Society for Vascular Surgery (SVS) bylaw referendum proposes changing the name attached to the membership status of Senior members as part of...
How a group of vascular surgeons is working to plug gaps in knowledge and inspire colleagues to recognize the specialty’s essential role in future...
The randomized data foundation on which clinicians base decision-making for vascular surgery patients is derived from populations that do not represent the patients they...
While a recent survey found that a majority of those who responded had performed mini-incision carotid endarterectomy (CEA) at least once in their practice...
The use of a paclitaxel-eluting stent to treat femoropopliteal occlusive disease led to equivalent outcomes in male and female patients in “a diverse and...
In this issue:
Wound healing in space: How a group of vascular surgeons is working to plug gaps in knowledge and inspire colleagues...
A retrospective, nationwide cohort study has revealed carotid endarterectomy (CEA) performed better than carotid artery stenting (CAS) in France over the course of a...
Imperative Care has announced the completion of patient enrollment in its SYMPHONY-PE study, a pivotal investigational device exemption (IDE) trial evaluating the safety and...
Envveno Medical has announced that its manuscript titled, "Three-year outcomes of surgical implantation of a novel bioprosthetic valve for the treatment of deep venous...
Endospan recently announced the presentation of 30-day results from the statistical dissection primary arm of the TRIOMPHE investigational device exemption (IDE) clinical study. The...
VVT Medical recently announced the appointment of Antonios Gasparis, MD, to its medical advisory board.
A press release notes that Gasparis brings over two decades...
Inquis Medical recently announced results from its AVENTUS trial evaluating the safety and efficacy of the company’s Aventus thrombectomy system.
The results were presented by...
The Society for Vascular Surgery (SVS), in collaboration with the Society for Vascular Nursing (SVN) and the American Podiatric Medical Association (APMA), has postponed...
The SVS Young Surgeons Section (YSS) has selected Nathan Liang, MD, of the University of Pittsburgh, to receive its 2025 visiting professorship.
Liang will...
The Society for Vascular Surgery (SVS) is inviting users to participate in a brief survey regarding their experiences with the sixth edition of the...
The Society for Vascular Surgery (SVS) will hold a virtual Highway to Health informational session exclusively for members on Wednesday, April 30, at 7...
The upcoming Vascular Annual Meeting (VAM), taking place June 4–7 in New Orleans, will see Jason T. Lee, MD, and Claudie Sheahan, MD, at...
In 2018, a group of dedicated vascular surgery trainees recognized a notable gap in educational resources within their specialty. This realization sparked the creation...
A new propensity-matched analysis of more than 1,100 lower extremity revascularization procedures carried out in patients with chronic limb-threatening ischemia (CLTI)—which aimed to help...
“We can almost be considered a specialty of colonizers, acquiring borrowed techniques. Why, then, do we see ourselves as the keepers of vascular care?...
“Where have all the doctors gone?” is the headline on the cover article of the January/February 2025 edition of the AARP Bulletin. Perhaps it...
This advertorial is sponsored by Penumbra, Inc.
Daniel Clair, MD, reflects on the STRIDE study of 30-day outcomes in patients who undergo first-line use of...
The established “gold-standard” approach to carotid revascularization remained enshrined as the go-to operation as far as a 2025 Charing Cross (CX) International Symposium audience...
R3 Vascular recently announced that the first patient in its ELITE-BTK pivotal trial has been treated by Brian DeRubertis, chief of vascular surgery at...
Jim Craig, a longtime support and vascular advocate, will deliver the annual keynote address at the 2025 Vascular Annual Meeting (VAM) in New Orleans...
Twelve-month outcomes of the VenaSeal Spectrum venous leg ulcer (VLU) trial, assessing time to ulcer healing following treatment with the VenaSeal (Medtronic) closure system,...
Results of the ACCESS 2 study, evaluating arteriovenous fistula (AVF) outcomes following use of the Sirogen (Vascular Therapies) sirolimus-eluting collagen implant, have shown that...
The next spring forward in aortic stent grafting will be rooted in a greater understanding of the biomechanical forces acting upon devices alongside continued...
In this issue:
The question of Vascular Surgery Board independence: Federated or free-standing
From Brazil to Baylor: Gustavo Oderich lands at cardiovascular powerhouse
...
Vascular and endovascular surgeons are frequently the only specialists other than primary care involved in the longitudinal care of their patients afflicted with clinically...
Venova Medical has announced the completion of patient enrollment in the company's VENOS-2 early feasibility study of the Velocity percutaneous arteriovenous fistula (pAVF) system...
The SVS International Relations Committee (IRC) has announced the recipients of the 2025 International Scholars Program, recognizing the achievements of three distinguished professionals: Sohei...
The Society for Vascular Surgery (SVS) has launched a search for a new editor-in-chief (EIC) for its flagship publication, the Journal of Vascular Surgery...
Following in the footsteps of some of the pioneers of cardiovascular surgery, Gustavo Oderich, MD, recently stepped into position as the Michael E. DeBakey...
A survey recently went out to Society for Vascular Surgery (SVS) members asking them to contribute their voice to an opinion poll over whether...
The 40-year history of endovascular aneurysm repair (EVAR) of the aorta wends a circuitous route, from behind the Iron Curtain in Soviet Ukraine, to...
Terumo Neuro has announced that its carotid stent system has received premarket approval (PMA) from the Food and Drug Administration (FDA).
This milestone marks the...
A new subanalysis of the SAVVE (Surgical antireflux venous valve endoprosthesis) trial found that there was no difference in the level of improvement in...
Cagent Vascular has announced its first patient enrolment in the Serranator versus plain balloon angioplasty optical coherence tomography (OCT) study.
This prospective, randomized (2:1 treatment...
Vincent Rowe, MD, from the University of California, Los Angeles (UCLA), became the 2025–26 Society for Clinical Vascular Surgery (SCVS) president during the Society’s...
The Vascular Research Initiatives Conference (VRIC) will spotlight the crucial role of mentorship in advancing the fields of vascular surgery and research during its...
The Society for Vascular Surgery (SVS) has launched registration for the sixth cohort of its Leadership Development Program (LDP). The curriculum is drawn from...
The Society for Vascular Surgery (SVS), in collaboration with the Society for Vascular Nursing (SVN) and the American Podiatric Medical Association (APMA), has designed...
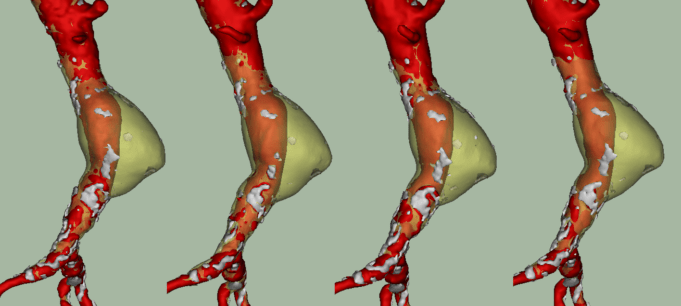
Inspection of aortic lumen, wall and thrombus volume changes throughout the cardiac cycle enables novel classification of abdominal aortic aneurysms (AAAs) into four types...
Surmodics has announced the commercial release of the Pounce XL thrombectomy system for endovascular removal of acute or chronic clot from peripheral arteries.
Intended for removal...
Gore recently announced the expansion of the Gore Tag conformable thoracic stent graft with Active Control system product line, following Food and Drug Administration...
Use of the Gore Tag thoracic branch endoprosthesis (TBE) “offers a safer alternative” to traditional thoracic endovascular aortic repair (TEVAR) with left subclavian artery...
Semaglutide significantly improved maximal walking distance in people with symptomatic peripheral arterial disease (PAD) and type 2 diabetes in a trial designed to evaluate...
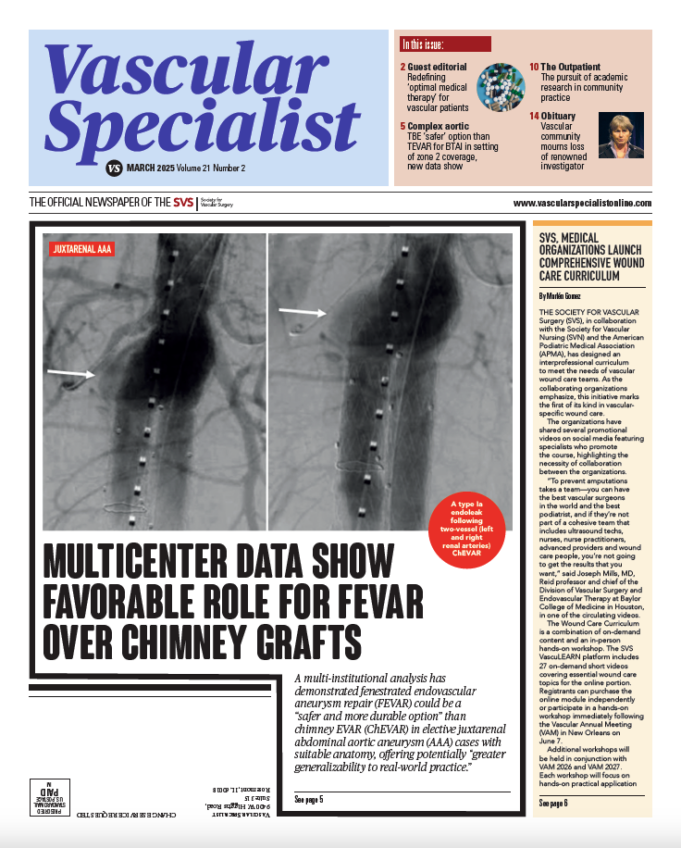
A multi-institutional analysis has demonstrated fenestrated endovascular aneurysm repair (FEVAR) could be a “safer and more durable option” than chimney EVAR (ChEVAR) in elective...
In this issue:
Multicenter data show favorable role for FEVAR over chimney grafts
SVS, medical organizations launch comprehensive Wound Care Curriculum
Guest editorial: Redefining...
Merit Medical Systems has announced that the six-month results from the randomized arm of the Wrapsody arteriovenous access efficacy—WAVE—trial are scheduled for publication in...
The importance of radiation safety training for vascular surgery trainees was highlighted by a recent study that detailed a learning curve effect between first-...
Imperative Care today announced Food and Drug Administration (FDA) 510(k) clearance of the 82cm version of its Symphony 16F catheter, the company’s latest innovation...
The Society for Vascular Surgery (SVS) has developed a comprehensive member toolkit to equip healthcare professionals, particularly vascular surgeons, with essential resources through its...
The Society for Vascular Surgery (SVS) will host a webinar titled "Harnessing AI in Vascular Surgery: Leading the Next Frontier" on March 27 from...
Addressing the audience at the 28th European Vascular Course (EVC 2025) in Maastricht, The Netherlands (March 9–11), Ulka Sachdev-Ost, MD, from the University of...
Th e Society for Vascular Surgery (SVS) Foundation, in partnership with the SVS Young Surgeons Section, has launched the VOYAGE Scholarship Program. This initiative will...
SonoVascular has announced the successful completion of an initial set of eight deep vein thrombosis (DVT) cases using its SonoThrombectomy system as part of...
Dennis Gable, MD, from The Heart Hospital Baylor Plano in Plano, Texas, discusses the unique imprint of the five-year follow-up from the GREAT registry...
Outgoing Vascular & Endovascular Surgery Society (VESS) President Misty Humphries, MD, used her presidential address to outline how trainees and young vascular surgeons can...
The SVS Foundation has various initiatives in place to celebrate Women’s History Month, including member profiles and patient-focused facts. New this year is fundraising...
The Society for Vascular Surgery (SVS) has opened its 2025 call for volunteers to serve on committees. In the member announcement that went out...
A new cross-sectional analysis as part of the SECOND trial found that most vascular surgery trainees are satisfied with their operative and clinical autonomy...
Dennis Gable, MD, from The Heart Hospital Baylor Plano in Plano, Texas, principal investigator for the thoracic component of the GREAT registry, explains that...
Janet Powell, the clinical trial investigator who played a pivotal role in several landmark abdominal aortic aneurysm (AAA) studies, has died at the...
As the new administration headed up by President Donald Trump gets underway, work has begun on his vision for a fundamental transformation of the...
At the end of my intern year, I was tired. It had been a long year of rounding, placing orders, seeing consults and refining...
Inari Medical, now part of Stryker, recently announced the launch of its Artix thrombectomy system. Purpose-built for the needs of the peripheral arterial system,...
When it comes to a presidential administration’s impact on our healthcare system, personnel is policy. So, a presidential transition is always a critical juncture...
Registration for the 2025 Vascular Annual Meeting (VAM 2025) is now open. This year’s meeting will take place in New Orleans, Louisiana, June 4–7.
VAM...
Philips has recently announced that it will no longer sell its Tack endovascular system in the U.S. following a Class I recall issued by...
In the second of a two-part series of editorials, Vascular Specialist Medical editor Malachi Sheahan III, MD, offers further insight into the world of...
Stereotaxis has announced a Food and Drug Administration (FDA) regulatory submission for the first robotically navigated catheter designed to expand usage of robotic magnetic...
Today, Shockwave Medical announced the U.S. launch of its Shockwave Javelin peripheral intravascular lithotripsy (IVL) catheter, a platform designed to modify calcium and cross...
“Battlefield first aid” after a colleague has a bad case as the cornerstone of peer-to-peer support in vascular surgery was the feature of the...
It is now common to see headlines like “When hospital prices go up, local economies take a hit” (Wall Street Journal), “Hidden hospital prices...
Ruth Bush, MD, used her 2025 American Venous Forum (AVF) presidential address to highlight the organization’s elevation and path to “level 5 leadership.”
Level 5...
Support from the Society for Vascular Surgery (SVS) and, later, funding from the SVS Foundation were integral to the BEST-CLI trial, said one of...
The 2025 Southern Association for Vascular Surgery (SAVS) presidential address featured the hashtag phrase #OnlyUs, an allusion to the specialty’s “unique” differentiator.
SAVS 2024–25 President...
The Society for Vascular Surgery (SVS) takes a moment to recognize the dedication, achievements and progress made over the past year. In a letter...
This advertorial is sponsored by AOTI.
Two prominent vascular surgeons discuss a rising tide of evidence pointing toward benefits of adjunct use of cyclically pressurized...
This advertorial is sponsored by Siemens.
Two leaders in vascular surgery preview the future of interventional imaging and consider the emergence of the hybrid operating...
The need for a revolutionary second generation of venous stents are among the gaps and unmet needs currently at play in deep venous surgery,...
The introduction of ketamine into a spinal cord ischemia (SCI) protection protocol used during thoracoabdominal aortic aneurysm (TAAA) repairs led to significant reductions in...
Preoperative diagnosis of lesion-specific ischemia using coronary computed tomography (CT)-derived fractional flow reserve (FFRCT) and ischemia-targeted coronary revascularization after carotid endarterectomy (CEA) can reduce...
Humacyte today announced the commercial launch of Symvess (acellular tissue engineered vessel-tyod) for use in adults as a vascular conduit for extremity arterial injury...
With the aims of predicting and comparing venous stent outcomes, aiding in communication with patients, and enhancing therapeutic decision-making, researchers have proposed an anatomical...
A multisite hospital system analysis of the safety and procedural learning curve behind the performance of percutaneous mechanical thrombectomy for pulmonary embolism (PE) shows...
In this issue:
US ARC uncovers ‘dramatic’ decrease in use of prophylactic spinal drains at same time as spinal cord ischemia steeply declines
Interview:...
Here's a closer look at a funded partially through grants from the Society for Vascular Surgery (SVS) Foundation.
Samantha Minc, MD, received a Clinical Research...
Shape Memory Medical today announced the first European enrollment in the AAA-SHAPE pivotal trial, the company’s prospective, multicenter, randomized, open-label trial to determine the...
Stryker announced today that it has completed the acquisition of Inari Medical. A press release notes that the addition of Inari brings an established...
Just over a week ago, I was racing my medical school vascular surgery attending, Dr. Adam Doyle, down a Breckenridge run memorably named “The...
Textbook outcomes remain “very rare” after revascularization for chronic limb-threatening ischemia (CLTI), with the main driver being an enduring inability to attain complete wound...
Society for Vascular Surgery (SVS) members have the opportunity to give and receive career guidance by participating in the Society’s Mentor Match program.
The SVS...
The 2025 Vascular Annual Meeting (VAM) will take place in New Orleans, Louisiana, from June 4–7, with a particular focus this year on innovation.
Key...
A new analysis of the BEST-CLI trial has produced the first evidence-based glomerular filtration rate (eGFR) value—30 mL/min/1.73 m2—as the cutoff below which patients...
These remain early days for transcatheter arterialization of the deep veins (TADV) in the setting of patients with severe chronic limb-threatening ischemia (CLTI), says...
InspireMD and North American Science Associates (NAMSA) today announced that, pursuant to a previously announced strategic outsourcing partnership, the companies are working together to conduct...
Akura Medical has announced today the first patient enrolment in the QUADRA-PE study evaluating the Katana thrombectomy system in patients with acute pulmonary embolism...
Thirty-day results from the ROADSTER 3 study have demonstrated that transcarotid artery revascularization (TCAR) is a safe and effective approach in patients who are...
Penumbra recently announced the launch of its Element vascular access system. This is the first laser-cut hypotube sheath designed for venous thromboembolism (VTE), the...
The Society for Vascular Surgery (SVS) announced that the Vascular Surgery MIPS Value Pathway (MVP) was posted for public comment by the Centers for...
With nomination deadlines now past, Section on Ambulatory Vascular Care (SAVC) award season is upon us, and past recipients of the Society for Vascular...
Despite a “dramatic” decrease in the use of prophylactic cerebrospinal fluid drains (CSFDs) for the prevention of spinal cord ischemia (SCI) in patients undergoing...
Intermountain Health St. Mary’s Regional Hospital in Grand Junction Colorado was awarded ACS/SVS Vascular Verification Program (Vascular-VP) designation on Nov. 19, 2024. By achieving...
Physicians are facing a fifth consecutive year of Medicare payment reductions due to policy adjustments and budget neutrality requirements in the Medicare Physician Fee...
A systematic review and meta-analysis of over 1,500 venous stenting procedures—said to be the first study on this topic to date—has highlighted an 18%...
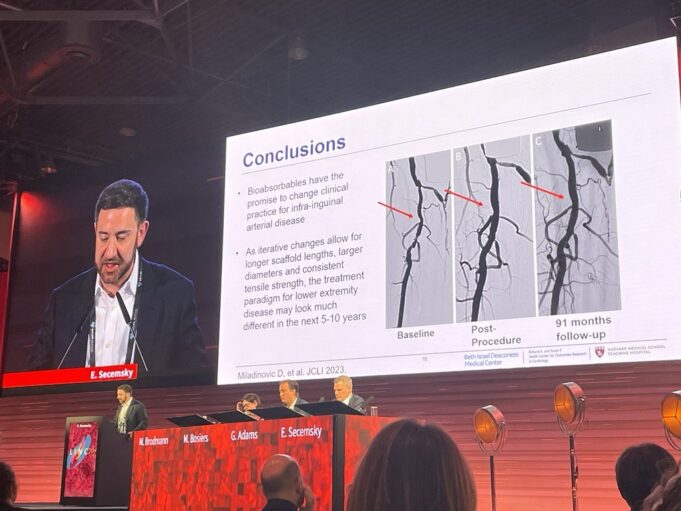
Pending the results of several trials and “iterative changes” to device design, bioabsorbable scaffolds are set to change the treatment paradigm for lower extremity...
The Society for Vascular Surgery’s (SVS) Young Surgeon Section (YSS) will close applications for its inaugural visiting professorship on Jan. 31. This scholarship program...
Cagent Vascular has announced the start of the POINT FORCE registry, a postmarket clinical follow-up study of the Serranator percutaneous transluminal angioplasty (PTA) serration...
Gore recently announced the first commercial use of its Excluder conformable abdominal aortic aneurysm (AAA) endoprosthesis with Active Control system in Canada.
The news coincides...
Margaret (Megan) Tracci, MD, has become the first-ever woman president of the Southern Association for Vascular Surgery (SAVS). The University of Virginia professor of...
The Food and Drug Administration (FDA) has issued a statement outlining measures to enhance protections against medical device shortages.
According to Michelle Tarver, director...
All those steps logged during the SVS Foundation’s exercise challenge in September will help the Foundation—and researchers—take new steps to unlock new treatments for...
Five prominent vascular surgeons have been named “at-large” members of the SVS Executive Board (EB), which has been newly restructured and expanded to increase...
The European Society for Vascular Surgery (ESVS) has developed new guidelines for the care of patients with vascular trauma. The 2025 clinical practice guideline...
A recent prospective, multicentre cohort study provides insights into early and late aortic-related mortality and rupture after fenestrated and branched endovascular aneurysm repair (F/BEVAR)...
Gore has announced recent CE mark of a lower profile Viabahn VBX balloon expandable endoprosthesis (VBX stent graft).
Medical Device Regulation (MDR) approval of this...
The one-year data from the SAVVE trial represent the “most encouraging clinical data that have ever been produced for a bioprosthetic deep vein valve”...
Researchers in Canada have developed machine learning (ML) models that they claim, “can accurately predict one-year IVC filter complications, performing better than logistic...
Boston Scientific has announced it has entered into a definitive agreement to acquire Bolt Medical, the developer of an intravascular lithotripsy (IVL) advanced laser-based...
A new Vascular Quality Initiative (VQI) data analysis, recently published in the European Journal of Vascular and Endovascular Surgery (EJVES), has found no statistically...
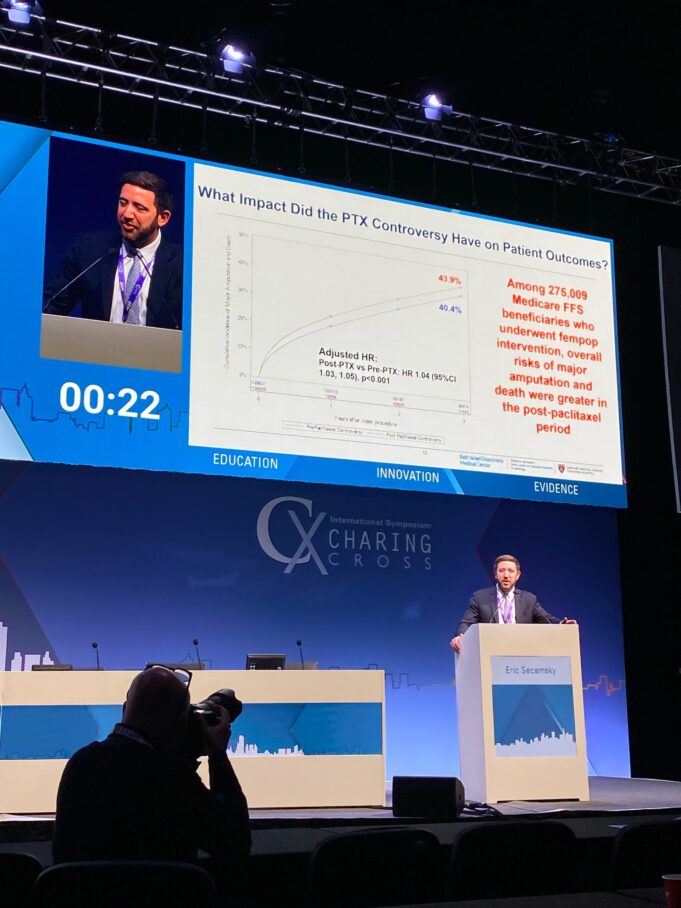
A recent analysis of over 270,000 Medicare fee-for-service beneficiaries has found an increase in adverse outcomes and death after a US Food and Drug...
A systematic review of U.S. literature focused on analyzing the effects of mergers and acquisitions on healthcare suggests that there is a lack of...
Ongoing studies of the Efemoral vascular scaffold system (EVSS)—previously described as a potential new paradigm in the treatment of long occlusive lesions—show that the...
I met my wife, Claudie, at the Harvard Medical School vascular research lab. This was not a romantic environment by most standards (depending, I...
The key role played by physician assistants (PAs) as part of vascular surgery teams in the U.S. came into focus during the Vascular Societies’...
Current graduates from both vascular surgery residency and fellowship programs are well prepared for surgical practice in aortic disease whether they are performing open...
Jean Panneton, MD, considers himself what he terms a “blue-collar” vascular surgeon as much as one engaged in academic practice and the necessity of...
This advertorial is sponsored by Inari Medical.
For Joseph M. Liechty, MD, a vascular surgeon with Texas Vascular Associates in Plano, Texas, the emergence of...
In this issue:
Positive one-year data for new venous valve portends promising future in chronic venous insufficiency treatment
Vascular trainees better prepared for open...
Humacyte recently shared that the Food and Drug Administration (FDA) has granted a full approval for the Symvess acellular tissue-engineered vessel.
A press release notes...
Merit Medical Systems announced today that the Wrapsody cell-impermeable endoprosthesis has received premarket approval from the US Food and Drug Administration (FDA). With this...
SAVVE trial principal investigators discuss results showing 98.4% VenoValve device patency, 85% clinically meaningful benefit and an 80% rate of ulcer size reduction at...
Ellen Dillavou, MD, division chief of vascular surgery at WakeMed Heart Center in Raleigh, North Carolina, primary investigator (PI) for the VenoStent trial, presented...
As part of a 2024 VEITHsymposium (New York City; Nov. 19–23) debate, Robert E Lee, MD, a medical officer recently retired from the Food...
At the recent VEITHsymposium (Nov. 19–23) in New York City, Marc Passman, MD, reflected on the achievements of the Society for Vascular Surgery (SVS)...
A rupture prediction-based algorithm is set to enhance patient selection for abdominal aortic aneurysm (AAA) repair. This is according to Randy Moore, MD, a...
This advertorial is sponsored by Tampa General Hospital.
As the prevalence of peripheral arterial disease (PAD) continues to climb across the U.S. and globally, a...
Ten-year data from the randomized HELP trial show that, while both endothermal ablation and conventional surgery are effective treatments for great saphenous varicose veins...
In residency and fellowships, vascular surgeons learn the techniques and skills to help them diagnose, manage and treat vascular disease.
But there are still important...
Vascular surgeon-scientists may submit their research for presentation at the 2025 Vascular Annual Meeting (VAM) in June through Wednesday, Jan. 8, 2025. The submission...
This month, Vascular Specialist launches its newest column, The Outpatient, which seeks to cover issues near and dear to the hearts of vascular surgeons...
Post election and with the end of the year on the horizon, a crucial but often overlooked period in the legislative calendar begins: the...
Vascular Specialist attended the SVS Complex Peripheral Vascular Interventions (CPVI) Skills Course and spoke with participants about the hands-on learning experience designed for vascular...
Biotronik today announced that it has concluded the BIO-OSCAR SOC study evaluating the baseline against which to measure the Oscar multifunctional catheter in treating complex...
The SVS Foundation is accepting applications for the 2025 James S.T. Yao Resident Research Award, which supports emerging medical professionals researching vascular disease biology...
When Frank Davis, MD, attended the SVS Vascular Research Initiatives Conference (VRIC) for the first time in 2014, he found not only meaningful discussions...
Microbot Medical recently announced that it has submitted a 510(k) premarket notification to the Food and Drug Administration (FDA) for its Liberty endovascular robotic...
I’m sure research residents everywhere have heard their fair share of unsolicited advice on this topic. But among all the voices advising me on...
Artivion recently announced that the Food and Drug Administration (FDA) has granted a humanitarian device exemption (HDE) for use of the AMDS hybrid prosthesis...
The Society for Vascular Surgery is seeking nominations for its annual Excellence in Community Practice Award.
Formerly known as the Excellence in Community Service Award,...
InspireMD today announced that the first patient has been enrolled in the company’s CGUARDIANS II clinical trial evaluating its CGuard Prime carotid stent system in...
A debate at this year's European Society for Vascular Surgery (ESVS 2024) annual meeting (Sept. 24–27) in Kraków, Poland, centered on innovation in vascular...
If you were in the audience at the 2024 Southern Association for Vascular Surgery (SAVS) annual meeting, you might have been taken aback by...
The Society for Vascular Surgery (SVS) has launched a new patient-facing website aimed at bridging the information gap between members of the public and...
The advent of large language models such as ChatGPT (Open AI) has left little of the world untouched—and a quintet of University of Toronto...
Pioneering American Venous Forum (AVF) and Eastern Vascular Society (EVS) efforts aimed at breaking down barriers to care and educating patients in economically challenged...
One in five patients failed to achieve a textbook outcome after undergoing carotid endarterectomy (CEA), transcarotid artery revascularization (TCAR) or transfemoral carotid artery stenting...
Bentley has announced the successful first-in-human implantation of its recently CE-certified BeFlared stent graft system, which the company states is the world’s first dedicated...
Newly released six-month results from single-arm arteriovenous graft (AVG) cohort in the WAVE (Wrapsody arteriovenous access efficacy) trial showed target lesion and access circuit...
Diabetes affects approximately 70% of patients with chronic limb-threatening ischemia (CLTI) and is associated with more severe presentation, driven mostly by higher rates of...
Latest study out of consortium details trends and outcomes after F/BEVAR procedures carried out over an eight-year period under consortium of 10 physician-sponsored IDEs.
Despite...
This advertorial is sponsored by Shockwave Medical.
Anand Prasad, MD, an 18-year veteran of vascular and endovascular medicine with a particular interest in peripheral arterial...
This advertorial is sponsored by Penumbra, Inc.
Adam Reichard, MD, discusses the use of Lightning Bolt 7® computer-assisted vacuum thrombectomy (CAVTTM) as a first-line option...
In this issue:
Service: Giving back—The Eastern Vascular Society (EVS) and American Venous Forum (AVF) pioneer vascular days of service in the community
...
Humacyte today announced the publication of clinical results evaluating the efficacy and safety of the acellular tissue engineered vessel (ATEV) in the repair of...
Envveno Medical has announced that it will present one-year data on all patients from the VenoValve US pivotal trial today at the VEITHsymposium (19–23 November, New York,...
enVVeno Medical today announced it has submitted an application with the U.S. Food and Drug Administration (FDA) seeking approval to market the VenoValve—a surgical...
A new analysis has found arm vein to be the most suitable alternative conduit to great saphenous vein (GSV) in chronic limb-threatening ischemia (CLTI)...
The sixth edition of the Vascular Education and Self-Assessment Program (VESAP6) is available for both members and nonmembers of the SVS to purchase. The...
The SVS Foundation “Voices of Vascular” series is celebrating Lyssa Ochoa, MD, for this year’s Hispanic History Month.
A Mexican American vascular surgeon, Ochoa is...
The Journal of Vascular Surgery-Cases, Innovations and Techniques (JVS-CIT) has appointed Juan Carlos Jimenez, MD, as its new associate editor and diversity, equity and...
On Tuesday, Sept. 24, The Society for Vascular Nursing (SVN), in collaboration with the PAD Collaborative and the American Association of Wound Care...
A webinar on “Perioperative Care in Open Aortic Vascular Surgery: Recommendations from ERAS and SVS” was held by the SVS Leadership Development Committee on...
The SVS and the American Pediatric Surgical Association (APSA) Pediatric Vascular Surgery Interest Group convened for its quarterly meeting on Tuesday, Oct. 1. The...
In collaboration with the Physician Assistant Section, the SVS Leadership Development Committee, is set to launch a leadership development podcast in partnerhsip with Audible...
The Vascular Quality Initiative (VQI) Patient Safety Organization’s Fellowship in Training (FIT) Program has announced its third class of FIT participants for 2024–25 alongside...
Abstracts for the 2025 Vascular Research Initiatives Conference (VRIC) are being accepted through Nov. 20.
VRIC, which is held in Baltimore, Maryland, this year, emphasizes...
Brajesh K. Lal, MD, assumed the Eastern Vascular Society (EVS) presidency during the 2024 EVS annual meeting. He took over from Kathleen Ozsvath, MD.
Patrick...
The Society for Vascular Surgery (SVS) has welcomed 11 surgeons to its ranks of Distinguished Fellows. The new fellows have distinguished themselves by making...
Findings from a retrospective analysis delivered at the recent European Society for Vascular Surgery (ESVS) annual meeting (Sept. 24–27) in Kraków, Poland, provide evidence...
In October, the most read stories from Vascular Specialist included a new leadership announcement from the American College of Surgeons (ACS); study results examining a...
Endologix has announced the final 36-month results from the DETOUR2 study—a prospective, single-arm, international, multicentre clinical evaluation of the novel Detour system for fully...
Penumbra has announced new data that demonstrate patients with intermediate-risk pulmonary embolism (PE) treated with Penumbra's computer-assisted vacuum thrombectomy (CAVT) technology have a shorter...
Shockwave Medical, part of Johnson & Johnson MedTech, has announced the first clinical outcomes associated with the Shockwave Javelin peripheral intravascular lithotripsy (IVL) catheter,...
Bolt Medical has announced the completion and results of the RESTORE ATK and RESTORE BTK pivotal clinical trials investigating the company's Bolt intravascular lithotripsy...
Presented today, late-breaking data from the second year of the LIFE-BTK clinical trial demonstrate the long-term effectiveness of the US Food and Drug Administration...
Royal Philips has announced enrolment of the first patient in the US THOR IDE clinical trial, which will study an innovative combined laser atherectomy...
R3 Vascular today announced that the Food and Drug Administration (FDA) has granted investigational device exemption (IDE) approval to initiate its ELITE-BTK pivotal trial...
A retrospective economic analysis of the LIFE-BTK trial has demonstrated the one-year cost-effectiveness of an everolimus-eluting resorbable scaffold over angioplasty for the treatment of...
The Centers for Medicare and Medicaid Services (CMS) has granted transitional pass-through (TPT) payment for Medtronic's Symplicity Spyral renal denervation (RDN) catheter and Recor...
The SVS Diversity, Equity and Inclusion (DEI) Committee continues to make strides in advancing inclusivity within the organization, with several projects underway to address...
Hospitals represent one of the largest industries in the U.S., with revenues greater than $1.4 trillion. The American Hospital Association reports that there are...
In the latest Corner Stitch column, medical student Bailey Richardson recounts the deeply personal life events that led to her unique journey into vascular...
Conflicting results open fresh debate around effect of vein harvesting technique on long-term patency of lower extremity bypass grafts.
Two separate analyses that looked at...
Vascular surgeon-scientists may submit their research for presentation at the 2025 Vascular Annual Meeting (VAM) in June starting from Nov. 6 through Wednesday, Jan....
The American College of Surgeons (ACS) elected Anton N. Sidawy, MD, as president-elect during the ACS annual business meeting on Oct. 22. He served...
Surmodics has announced that early results of a subset of 60 real-world acute, subacute, and chronic limb ischaemia patients from its PROWL registry study...
The Japanese concept of ikigai, or an individual’s reason for being, underpinned the message behind a fresh call for mentorship in vascular surgery and...
Findings from the first international randomised controlled trial (RCT) to compare patient outcomes following treatment with large-bore mechanical thrombectomy (LBMT) versus catheter-directed thrombolysis (CDT)...
Data from the prospective, multicentre, investigator-initiated SIRONA randomised clinical trial demonstrated MagicTouch (Concept Medical) sirolimus-coated balloons to be noninferior compared to paclitaxel-coated balloons with...
Jessica P. Simons, MD, has become the 2024–25 president of the New England Society for Vascular Surgery (NESVS). She is the first woman to...
Vivasure Medical has announced initial positive results from its US IDE PATCH pivotal study evaluating the safety and efficacy of the Vivasure PerQseal closure...
Thrombolex has announced the enrolment of the first two patients in the RAPID-PE study using the Bashir endovascular catheter for the treatment of acute...
In his address at the 2024 Vascular Annual Meeting (VAM), Society for Vascular Surgery Immediate Past President Joseph Mills, MD, eloquently stated—and please excuse...
According to a new national survey released by the Society for Vascular Surgery (SVS), nearly one in three Americans at the highest risk for developing...
Results from a subset analysis of the ABRE study have shown that patients in three out of the six World Health Organisation (WHO) body...
A new analysis that shows current smokers who undergo carotid endarterectomy (CEA) for asymptomatic carotid stenosis are at increased risk of long-term stroke and...
The American Board of Surgery (ABS) is accepting nominations for vascular surgeons to serve on its council for a six-year term, beginning on...
The Society for Vascular Surgery (SVS) online learning management system (LMS), now known as SVS VascuLEARN. The platform was formerly called SVS OnDemand. VascuLEARN...
Mid-term results from an analysis of a series of hybrid aortic arch reconstructions utilizing overlapping single thoracic branch endoprostheses in a dual-branch configuration showed...
Results from a new study highlighting that female patients are at increased risk of adverse outcomes after acute limb ischemia (ALI) lend weight to...
An observational study of intermittent claudication (IC) practice patterns before and after publication of the Society for Vascular Surgery (SVS) appropriate use criteria (AUC)...
Endospan today announced the completion of enrolment for the primary arm of its TRIOMPHE investigational device exemption (IDE) clinical study investigating the Nexus aortic...
CVRx, the company behind the Barostim baroreflex activation therapy system, recently announced that the American Medical Association (AMA) has accepted new Current Procedural Terminology...
A prominent surgeon-scientist and Society for Vascular Surgery (SVS) member is the 2024 recipient of the American Heart Association (AHA) Joseph A. Vita Award,...
Pilot testing has shown that a new framework—dubbed Endo-STAR—can be used to describe and standardise endovascular interventions for peripheral arterial disease (PAD) within a...
This advertorial is sponsored by Shockwave Medical.
Paul J. Foley III, MD, a vascular surgeon and director of the non-invasive vascular lab at Doylestown Hospital...
This advertorial is sponsored by Surmodics.
Interventions for acute limb ischemia (ALI) represent up to 16% of the case volume for vascular surgeons and cost...
Amplitude Vascular Systems (AVS) recently announced that it has enrolled the first patient in its U.S. pivotal trial for pulsatile intravascular lithotripsy (IVL) therapy....
In this issue:
Endoscopic vein harvest under the microscope
Study details aspects of early care improvements after publication of SVS appropriate use criteria for...
Outgoing Midwestern Vascular Surgical Society (MVSS) President Bernadette Aulivola, MD, underscored the importance of “investing in the people of vascular surgery—those learning about it...
Contego Medical today announced the US Food and Drug Administration's (FDA) premarket approval (PMA) of the Neuroguard IEP system for carotid revascularisation.
The company shares...
This advertorial is sponsored by Inari Medical.
Despite the recent growth in mechanical thrombectomy devices for acute deep vein thrombosis (DVT), interventional options for wall...
In this supplement, sponsored by AngioDynamics:
The storied history behind the Auryon Atherectomy System
What micro-CT reveals about the effectiveness of the Auryon laser...
Published in the Journal of Thrombosis and Thrombolysis, new research with a follow-up of 27 years has found an association between high levels of...
A new paper that drills into the impact of a novel bioprosthetic venous valve replacement by CEAP (Clinical, Etiological, Anatomical and Pathophysiological) classification shows...
InspireMD has announced that the Food and Drug Administration (FDA) approval of the company’s investigational device exemption (IDE) application to initiate the CGUARDIANS II...
An analysis of the BEST-CLI randomized controlled trial (RCT) found that smokers with chronic limb-threatening ischemia (CLTI) should be preferentially treated with open bypass...
Outgoing Eastern Vascular Society (EVS) President Kathleen Ozsvath, MD, used her presidential swansong to address the needs of those practicing the specialty moving forward,...
Medinol has announced the successful first-in-human implantation of the ChampioNIR drug-eluting peripheral stent by Gerard S. Goh, MD, head of interventional radiology, and Thodur...
New study data suggest that the interventional treatment of symptomatic carotid artery disease may result in an improved rate of age-adjusted overall survival compared to medical...
Cook Medical this week announced that it has created a custom-made device for the frozen elephant trunk (FET) aortic procedure.
A press release details that,...
In September, the most read stories from Vascular Specialist included a change in leadership for the Midwestern Vascular Surgical Society; study results examining new treatment...
Surmodics has announced that it has received Food and Drug Administration (FDA) 510(k) clearance for its Pounce XL thrombectomy system.
The Pounce system is indicated...
A new deep vein thrombosis (DVT) diagnostic pathway incorporating non-expert artificial intelligence (AI)-guided compression ultrasound could reduce workload and costs for healthcare systems...
As Matthew Eagleton, MD, continues with the duties of president of the Society for Vascular Surgery (SVS), his vision for the organization extends well...
The Association of Program Directors in Vascular Surgery (APDVS) has released a new ebook designed to enhance vascular surgery education for medical students. According...
A foundational element for effective advocacy is understanding the profound impact that legislative decisions have on the practice of medicine and the health of...
Welcome to the CLTI Chronicles, a saga of mystery, medicine and misadventures in limb salvage unfolding weekly in my office, where the drama of...
The Society for Vascular Surgery (SVS) is encouraging members to participate in the 2024 Compensation Survey, a key initiative designed to provide vascular surgeons...
The Society for Vascular Surgery (SVS) Quality Improvement Committee has developed toolkits aimed at enhancing patient care and outcomes by integrating patient-reported outcomes (PROs)...
The Society for Vascular Surgery (SVS) has called om members to detail their educational and professional development needs in order that it can seek...
A large meta-analysis of elective endovascular aneurysm repair (EVAR) outcomes by age has shown that three-to-five-year survival is very low in octogenarians, prompting researchers...
Brajesh K. Lal, MD, assumed the Eastern Vascular Society (EVS) presidency during the 2024 EVS annual meeting in Charleston, South Carolina (Sept. 19–22).
The professor...
Boston Scientific announced the close of its acquisition of Silk Road Medical, the company behind transcarotid artery revascularization (TCAR).
"The integration of the TCAR platform into our portfolio...
The Society for Vascular Surgery (SVS) announced the merger of its Section on Outpatient and Office Vascular Care (SOOVC) and the Community Practice Section...
Ahmed Abou-Zamzam Jr., MD, has taken over the as the 2024–25 president of the Western Vascular Society (WVS).
The professor of surgery at Loma Linda...
Merit Medical has today announced positive six-month findings from the randomized arteriovenous (AV) fistula arm of its Wrapsody arteriovenous access efficacy (WAVE) pivotal trial....
Royal Philips today announced the introduction of the 160cm Food and Drug Administration (FDA)-approved version of its LumiGuide endovascular navigation wire. This enhanced LumiGuide...
Since there are more than 40 million Spanish speakers in the United States, training more bilingual physicians could potentially improve equitable care in Latino...
A Vascular Quality Initiative (VQI) analysis of evolving practice in the wake of the BEST-CLI trial demonstrated no increase in the rate of surgical...
SVS Executive Director Kenneth M. Slaw, PhD, examines the key forces and factors shaping vascular surgery and how they may interact to create different...
Patrick Muck, MD, assumed the reins of the Midwestern Vascular Surgical Society (MVSS) at the conclusion of the 2024 MVSS annual meeting in Chicago...
A pilot study in Canadian capital Ottawa aimed at uncovering the extent of undiagnosed vascular disease among the city’s homeless population detected a subset...
Argon Medical has today announced the launch of the Cleaner Vac thrombectomy system for the removal of blood clot from the peripheral venous vasculature.
The...
The Association of American Medical Colleges (AAMC) has elected Julie A. Freischlag, MD, as chair of its 2024–25 board of directors. Her term will...
Vascular Specialist is launching a new column in the October issue of the newspaper dedicated to issues important to—and involving—the office-based lab (OBL) setting.
Entitled...
Results from a single-center analysis show the outpatient setting met SVS objective performance goals.
A retrospective study of endovascular interventions performed for chronic limb-threatening ischemia...
In this issue:
‘Select CLTI patients may be safely treated in the office-based lab’: Single-center analysis finds outpatient setting met SVS objective performance goals
...
Shockwave Medical, part of Johnson & Johnson MedTech, has announced the full U.S. launch of its Shockwave E8 peripheral intravascular lithotripsy (IVL) catheter, following...
In August, the most read stories from Vascular Specialist included a research letter addressing ‘alarmingly high’ mortality rates in chronic limb-threatening ischemia (CLTI) patients; a new...
The Society for Vascular Surgery (SVS) Executive Board (EB) gathered at its headquarters in Rosemont, Illinois, this summer, tackling over 40 strategic, policy and...
In a recently conducted survey, over 200 U.S. advanced practice providers (APPs) in vascular surgery cited performing at the top of their license as...
In recognition of Vascular Nurses Week (Sept. 8-14), Vascular Specialist spoke with Society for Vascular Nursing (SVN) President Karen Bauer, DNP. Bauer is a...
NOTE: This video is ONLY available to watch in selected countries and geographies
Jean Bismuth, MD, the University of South Florida Health and Tampa General...
The Society for Vascular Surgery Vascular Quality Initiative (SVS VQI) clinical registry today announced the Carotid Care Quality Champion recognition program.
The program honors healthcare...
The 2024 SVS Foundation’s Vascular Health Step Challenge kicked off Sept. 1 with over 400 registrants committed to walking 60 miles throughout September in...
Koya Medical recently announced that full results from the TEAYS clinical study have been published in the Journal of Vascular Surgery (JVS).
The data...
VVT Medical recently announced that the American Venous Forum (AVF) has provided an official opinion letter regarding CPT coding guidance for the ScleroSafe percutaneous...
While the 2024 Vascular Annual Meeting (VAM) only wrapped up in June and attendees are still in awe of its education offerings, it is...
The SVS Executive Board (EB) convened on July 25 under the leadership of SVS President Matthew Eagleton, MD, to accelerate the implementation of key...
The SVS has called on its members to nominate individuals to fill five at-large positions on the Executive Board (EB) as required following a...
The SVS Foundation hosted its annual gala on June 21 at Chicago’s famous Museum of Science and Industry. This was the first of the...
The Society for Vascular Surgery (SVS) Executive Board (EB) unanimously approved the transition of the Young Surgeons Section (YSS) from a pilot section to...
The Society for Vascular Surgery’s (SVS) Advocacy team is thrilled to announce the inaugural Leadership and Advocacy Conference, set to take place Sept. 14–16,...
The Society for Vascular Surgery (SVS) has partnered with KarenZupko and Associates (KZA), a consulting and education firm, to create vascular surgery coding resources...
AngioDynamics today announced European CE mark approval of the Auryon atherectomy system, designed for the treatment of peripheral arterial disease (PAD), including chronic limb-threatening...
As my third year of residency comes to a close, that little thing called The Match feels like a distant fever dream. The...
Patients with peripheral arterial disease (PAD) reporting lower levels of social support experience worse health outcomes, a new Yale-led study finds.
Social support is thought...
While nobody died, the procedure could have gone smoother. The patient, a corpulent man in his late 40s had been on dialysis for half...
The European Society of Cardiology (ESC) has today published its 2024 guidelines for the management of peripheral arterial and aortic diseases (PAAD), evaluating these...
SonoVascular has announced the successful initiation of its first-in-human study (FIH) of the company's SonoThrombectomy system, an ultrasound-facilitated, thrombolytic-enhanced platform that utilizes multiple mechanisms...
The Food and Drug Administration (FDA) has issued a Class I recall—the most serious type—for Inari Medical's ClotTriever XL, 30mm device due to reports...
The Society for Clinical Vascular Surgery (SCVS) has issued a call for abstracts for its 2025 Annual Symposium, with just under two weeks left...
The Eastern Vascular Society (EVS) has incorporated a day of service into its 2024 annual meeting, being held in Charleston, South Carolina (Sept. 19–22).
Leading...
The recent Vascular Annual Meeting (VAM 2024) was more than a gathering of medical professionals to discuss advancements in vascular health. It featured the...
The Society for Vascular Surgery (SVS) announced an increase in membership dues for 2024 for Active members, a strategic move aimed at enhancing support...
Michael Conte, MD, recently used his 2024 Veith Lecture to call on young vascular surgeons on the peripheral arterial disease (PAD) frontlines to drive...
Researchers have reported a 94% technical success rate among other key findings from a recent international, multicentre, single-arm cohort study of physician-modified endografts (PMEGs)...
In this issue:
‘Choosing the right devices for the right patients’: Surgeon-scientist secures NIH funding to develop MRI technique for precision PAD care
SVS...
A new research letter underscores the need to improve long-term survival following lower extremity revascularization for chronic limb-threatening ischemia (CLTI), setting out a randomized...
The potential for cost and time to be key factors in low levels of outcomes data reporting emerged during a national survey of the...
The diagnosis and management of non-thrombotic iliac vein lesions (NIVL) is the focus of a newly published set of consensus statements from the Vascular...
The Society for Vascular Surgery (SVS) has expressed conditional support for cardiology establishing an independent certification board but objects to provisions in the current...
Centerline Biomedical recently announced that its Intra-Operative Positioning System (IOPS) Viewpoint catheter has received Food and Drug Administration (FDA) 510(k) clearance. The Viewpoint catheter is...
Vascular Therapies has today announced completion of enrollment in the ACCESS 2 trial, a Phase III prospective randomized, multicenter clinical study of Sirogen for...
The 2024 Vascular Annual Meeting (VAM) saw the culmination of a decade-long project by the Society for Vascular Surgery's (SVS) History Project Work Group....
Silk Road Medical has announced the milestone of more than 100,000 transcarotid artery revascularisation (TCAR) procedures having been performed to date.
“A growing number of...
The Cardiovascular and Interventional Society of Europe (CIRSE) have announced that patient enrolment for the CALCIO trial has begun. CALCIO started patient enrolment on...
Thrombolex and Aidoc have announced a strategic partnership aimed at revolutionising the treatment of acute pulmonary embolism (PE), a press release reports.
This strategic collaboration,...
Patients who undergo carotid revascularization at the weekend have increased odds of complications and mortality, with transfemoral carotid artery stenting (TFCAS) posing the highest...
Researchers at the University Medical Center Groningen (Groningen, The Netherlands) and the University of Twente (Enschede, The Netherlands) have developed a prediction model that...
One-year outcomes from the STRIDE study of the Indigo aspiration thrombectomy system (Penumbra) for lower-extremity acute limb ischemia (ALI) showed target limb salvage and...
Five-year outcomes the PRESERVE II pivotal trial of Cook Medical’s Zenith branch iliac graft (ZBIS) demonstrate “sustained safety and effectiveness” when combined with the...
On Friday, June 21, the Society for Vascular Surgery (SVS) Young Surgeons Section (YSS) held their session at the 2024 Vascular Annual Meeting (VAM)....
The Society for Vascular Surgery (SVS) convened its Annual Business Meeting at the Vascular Annual Meeting (VAM) on June 22 in Chicago.
A highlight...
“Life over limb”—that was one of the key messages to emerge from the VAM 2024 SVS Women’s Section session that placed a lens over...
The E. Stanley Crawford critical issues forum at VAM 2024 in Chicago—which featured the tagline ‘Quality Care Everywhere’—underscored the importance of standards, infrastructure, personnel...
As a vascular surgery resident, our training emphasizes anticipating and planning for complications. This mindset is undoubtably critical clinically but can be less beneficial...
Stakeholder organizations such as the Society for Vascular Surgery (SVS) play a crucial role in shaping healthcare policies and ensuring that the voices of...
This advertorial is sponsored by AOTI.
Until about a decade and a half ago, vascular surgeon Alisha Oropallo, MD, could be counted among the skeptics....
Humacyte recently announced it has been granted Regenerative Medicine Advanced Therapy (RMAT) designation from the US Food and Drug Administration (FDA) for its investigational...
Life Seal Vascular recently announced that it has been awarded a National Science Foundation (NSF) Small Business Innovation Research (SBIR) grant. The funding will...
Attendees of the 2024 Vascular Annual Meeting (VAM) can now access recordings of the sessions, including poster presentations, lectureships and more. The recordings are...
The Society for Vascular Surgery (SVS) Foundation has appointed new members to its Board of Directors. These individuals will continue the Foundation’s mission of...
Bhagwan Satiani, MD, explores the entrance of insurance companies into the physician practice acquisition market.
The well-written recent editorial from Mal Sheahan, Vascular Specialist medical...
Huge opportunities abound for vascular surgeons to take a leadership role on the frontlines of a developing peripheral arterial disease (PAD) treatment crisis in...
Many attending the 2024 Vascular Annual Meeting (VAM; June 19–22) might have been unaware of the early life of 2023–24 Society for Vascular Surgery...
In this issue:
Grasping the nettle—huge opportunity for vascular surgeons to take the lead on the frontlines of peripheral arterial disease (PAD) treatment crisis,...
In June, the most read stories from Vascular Specialist included key reports from the 2024 Vascular Annual Meeting (VAM; June 19–22) covering TEVAR, CEA...
Theraclion, a French MedTech company developing a robotic platform for non-invasive high-intensity focused ultrasound (HIFU) therapy, announces today that treatments in the Food and...
Humacyte recently announced it has been granted Regenerative Medicine Advanced Therapy (RMAT) designation from the US Food and Drug Administration (FDA) for its investigational...
Registration for the Society for Vascular Surgery (SVS) Foundation’s 2024 Vascular Health Step Challenge is now open. The Foundation is hosting the third annual...
Those looking for a comprehensive review resource for vascular surgery are encouraged to check out the Society for Vascular Surgery’s (SVS) sixth edition of...
New data presented at the 2024 Vascular Annual Meeting (VAM) held June 19–22 in Chicago have revealed poor overall rates of medical therapy adherence...
The Society for Vascular Surgery (SVS) has submitted comments to the Federal Trade Commission (FTC), the Department of Justice (DOJ) and the Department of...
Study findings were presented during Saturday, June 22’s Plenary Session 7 support current vascular guidelines advocating for effective supervised exercise therapy (SET) prior to...
Gore has announced the first commercial use of the Gore Tag thoracic branch endoprosthesis (TBE) in Canada.
The news came as Canadian government health authority...
The 2024 Vascular Annual Meeting (VAM) in Chicago showcased emerging talent in vascular surgery through a moderated Poster Competition held over two days of...
A recent study suggests that endovascular treatment of the common femoral artery (CFA) is associated with an increased rate of long-term CFA-specific reintervention, regardless...
The US Food and Drug Administration (FDA)-approved Gore Excluder Thoracoabdominal Branch Endoprosthesis (TAMBE) has been shown to be safe and effective at 30 days...
High stroke and death rates relating to carotid stenting in the Pacific Northwest may be driven by the selection of high-risk patients with less...
A study examining outcomes between different carotid revascularisation procedures has concluded that transcarotid artery revascularisation (TCAR) and carotid endarterectomy (CEA) both represent a “reasonable choice” in severe chronic...
The Society for Vascular Surgery (SVS) has announced Linda Harris, MD, as its next vice president, meaning that she enters the presidential line and...
Huge opportunities abound for vascular surgeons to take a leadership role on the frontlines of a developing peripheral arterial disease (PAD) treatment crisis in...
As the landscape of surgical education undergoes rapid evolution, one innovation promises to redefine how future vascular surgeons are trained and certified: Entrustable Professional...
The SVS sub-section on Outpatient and Office Vascular Care (SOOVC) Chair Anil Hingorani, MD, used the group’s dedicated VAM 2024 session to focus on...
A standing ovation, flashing lights and a praiseworthy introduction greeted the recipients of the SVS Lifetime Achievement Award yesterday morning: Peter Gloviczki, MD, and...
The SVS Women's Section will host their VAM 2024 session, “Vascular Trauma: What I Need to Know in the Middle of the Night” at...
Five-year outcomes from the PRESERVE II study support the safety and effectiveness of the investigational Zenith iliac branch graft (ZBIS, Cook Medical) in combination...
“Our findings ultimately suggest that vascular evaluation within six weeks of wound appearance can significantly supplement wound healing in wound centers,” said Saranya Sundaram,...
In this issue:
Chickasaw Nation study exemplifies how diversity can boost patient access to specialized vascular care
Young vascular surgeons on the PAD frontier:...
A new contemporary comparative study will be presented during Friday’s Plenary Session 5 (8–9:30 a.m., West Building, Level 3, Skyline Ballroom) by author Theodore...
SVS President Joseph Mills, MD, is set to issue a rallying call steeped in classical history and decades of collected vascular surgery wisdom in...
Daniel McDevitt, MD, spoke during the Community Practice Section at VAM 2024 on potential methods to participate in the shift to the office-based lab...
“Our findings ultimately suggest that vascular evaluation within six weeks of wound appearance can significantly supplement wound healing in wound centers,” said Saranya Sundaram,...
The International Chapter Forum Educational Session at VAM 2024 saw Ana M. Botero, MD, chief of vascular surgery at Fundación Santa Fe de Bogotá...
A “feasible and reproducible” intervention bundle characterized by surveys, provider education and patient screening has demonstrated the potential to not only identify gaps in...
“True diversity is a good and powerful thing that should be celebrated. It's not mandatory quotas; it's intentional inclusion," said Karith Foster, chief executive...
Transparency was at the forefront of a My Worst Cases session at VAM yesterday afternoon. Moderator William Robinson, MD, professor and chief in the...
"All vascular surgeons have access to the same randomized trials and guidelines, which should harmonize international treatment patterns, but this is not always the...
Clinical guidelines from the Society for Vascular Surgery (SVS) and the European Society for Vascular Surgery (ESVS) are pivotal in directing practitioners' management of...
Diabetes-related lower extremity amputations (LEA) disproportionately impact racial minorities and low-income groups. The Vascular Volunteers in Service to All (VISTA) assessment tool identified significant...
The society for vascular nursing (SVN) is in the midst of its 42nd Annual Conference at McCormick Place, West. The conference kicked off Wednesday...
A new secondary analysis of sex disparities in the best endovascular versus best surgical therapy in patients with critical limb ischemia (BEST-CLI) trial data...
Physician-sponsored Investigational Device Exemption (IDE) trials have an important role to play in the continued development and refinement of aortic endovascular devices, attendees of...
“I urge each one of you to seize this unique opportunity to not only enhance your patient care but also elevate your careers,” SVS...
On Friday morning, June 21 (6:30–7:15 a.m. in the West Building, Level 1), VAM 2024 attendees will have the chance to take in an...
In this issue:
Attendees welcomed to ‘more inclusive, more innovative, and more educational’ VAM
Endovascular CFA treatment associated with increased rate of long-term...
“If you had cancer you would go to a cancer center where you would get a multidisciplinary approach,” SVS President Joseph L. Mills, MD,...
A new secondary analysis of sex disparities in the best endovascular versus best surgical therapy in patients with critical limb ischemia (BEST-CLI) trial data...
A study examining outcomes between different carotid revascularization procedures has concluded that transcarotid artery revascularization (TCAR) and carotid endarterectomy (CEA) both represent a “reasonable...
High stroke and death rates relating to carotid stenting in the Pacific Northwest may be driven by the selection of high-risk patients with less...
Results of a sub-study from the pivotal Investigational Device Exemption (IDE) trial assessing Gore’s Excluder conformable abdominal aortic aneurysm (AAA) endoprosthesis have shown...
A growing prevalence of peripheral arterial disease (PAD) across the world parallels a deteriorating PAD landscape in the U.S., underscoring a need for more...
The SVS Community Practice Section (CPS) has put together an extensive 90-minute program for Thursday afternoon at VAM.
The session, entitled Community Practice Section: Optimizing...
The BEST-CLI trial underscores the importance of ensuring trainee competence in open peripheral revascularization as well as endovascular treatment. Indeed, the trial results have...
“Variability is a source of inferior quality, poor outcomes and harm,” says Anil Hingorani, MD, vascular surgeon at New York University (NYU) Langone in...
The Society for Vascular Nursing (SVN) is again hosting its 42nd Annual Conference June 19–20 at McCormick Place alongside VAM 2024. This year’s meeting...
New data presented during Friday's Plenary Session 6 (10–11 a.m. in the West Building, Skyline Ballroom) will shed light on trends in the adoption...
The advent of artificial intelligence (AI) in vascular surgery holds great promise to optimize the delivery of care, with prospective uses including patient selection...
The focus of yesterday’s American Pediatric Surgical Association (APSA)-SVS task force’s spring quarterly interest group meeting at VAM 2024 was the “incredibly crucial” topic...
A recent study suggests that endovascular treatment of the common femoral artery (CFA) is associated with an increased rate of long-term CFA-specific reintervention, regardless...
Selected research to be presented during the Wednesday afternoon, June 19, series of Vascular and Endovascular Surgery Society (VESS) Scientific Sessions (from 1:30 p.m....
If you had cancer you would go to a cancer center where you would get a multidisciplinary approach,” SVS President Joseph Mills, MD, tells...
During the first day of VAM 2024, the Society for Vascular Surgery (SVS) and American Venous Forum (AVF) have co-sponsored a session on the...
Contemporary management of carotid disease after the Centers for Medicare and Medicaid Services (CMS) carotid artery stenting (CAS) decision memo and prior to CREST-2...
A 2024 Vascular Annual Meeting (VAM 2024) Education Session taking place on Wednesday, June 19 (3:15–4:45 p.m. in the West Building, Level 1) will...
Some of the benefits to quality care provision ensuing from participation in the American College of Surgeons (ACS) and SVS Vascular Verification Program (Vascular-VP)...
To help us preview some of the sessions of interest I reached out to the chairs of the SVS Program and Postgraduate Education Committees—Andy...
In this issue:
The Crawford Forum: Quality in care is this year’s critical issue
National Coverage Determination: Data and discussion come at key moment...
This year’s James S. T. Yao Resident Research Award winner looks at novel therapeutic target for non-healing diabetic wounds. Kevin D. Mangum, MD, PhD,...
Against the backdrop of a national shortage in the vascular surgery workforce, a recent study has found that the Society for Vascular Surgery (SVS)...
The director of the Vascular Low Frequency Disease Consortium (VLFDC) is set to be among the headline presenters at this year’s World Federation of...
An education session set to take place in the afternoon of Wednesday, June 19 (3:15–4:45 p.m. in the West Building, Level 1), will examine...
In 2021, the Society for Vascular Surgery (SVS) and the American Pediatric Surgical Association (APSA) established a task force dedicated to optimizing pediatric vascular...
The Vascular Quality Initiative (VQI) is hosting its two-day annual conference at the 2024 Vascular Annual Meeting (VAM 2024) from June 18–19, drawing healthcare...
It is with great excitement and anticipation that I welcome you to the Vascular Annual Meeting (VAM), taking place from June 19–22 in the...
Boston Scientific has announced that it has entered into a definitive agreement to acquire Silk Road Medical, a medical device company involved in producing...
The Society for Vascular Surgery (SVS) and American College of Surgeons (ACS) Vascular Verification Program (Vascular-VP) will be a central focus on the opening...
This advertorial is sponsored by Laminate Medical Technologies.
At the 2024 Charing Cross (CX) International Symposium (April 23–25) in London, England, the Vascular Access Masterclass...
When it comes to the ongoing debate of carotid endarterectomy (CEA) versus transcarotid artery revascularization (TCAR) in the treatment of carotid artery stenosis, operators...
A key event on informed decision-making in chronic limb-threatening ischemia (CLTI) promises to be a highlight of the 2024 Vascular Annual Meeting (June 19–22). Funded...
The Society for Vascular Surgery (SVS) has officially launched registration for the annual Complex Peripheral Vascular Initiatives (CPVI) Skills Course. The first course launched...
The 2024 Venous Symposium (May 8–11) in New York City was highlighted by keynote speaker Peter Pappas, MD, regional medical director and program director...
Royal Philips today announced the first implant of the Duo venous stent system, indicated to treat symptomatic venous outflow obstruction in patients with chronic...
Amplitude Vascular Systems (AVS) announced today that it has received an investigational device exemption (IDE) from the Food and Drug Administration (FDA) to begin...
In this issue:
Verified vascular! Quality care in vascular disease will form a central backdrop to this year’s Vascular Annual Meeting (VAM)
VAM...
Sean P. Lyden, MD, chairman of the Department of Vascular Surgery at Cleveland Clinic’s Heart, Vascular and Thoracic Institute in Cleveland, Ohio, will discuss...
On Jan. 22, 2024, the American Heart Association (AHA) and the American College of Cardiology (ACC), together with three other major cardiology societies, submitted...
In April, the most read stories from Vascular Specialist include new data from the C-GUARDIANS pivotal investigational device exemption (IDE) trial presented at the...
During the 2024 Charing Cross (CX) International Symposium in London, England (April 23–25), a Great Debate that pitted four global peripheral arterial disease (PAD)...
Shape Memory Medical recently announced that the first patient has been treated in the AAA-SHAPE pivotal trial.
AAA-SHAPE (Abdominal aortic aneurysm sac healing and prevention...
At the Leipzig Interventional Course (LINC) 2024 (28–31 May, Leipzig, Germany), Michael Lichtenberg (Arnsberg Vascular Clinic, Arnsberg, Germany) shared for the first time a...
Patrick Muck, MD, from Cincinnati, Ohio, provided a state of the venous stent landscape during IVC 2024. The TriHealth vascular surgery program director ran...
The importance of activating the calf muscle pump in the treatment of lower extremity lymphedema came to the fore during the 2024 International Vein...
During the 2024 American Venous Forum (AVF;March 3–6) in Tampa, Florida, multiple presenters discussed inequities in venous care with a focus on how to...
Attending a conference such as the Vascular Annual Meeting (VAM) is a great way to increase step count and get blood flowing, which is...
"Every so often in vascular disease, disruptive technologies come along and the question really is, ‘Is this going to make a big impact on...
Money permeates every single aspect of American politics. To run a congressional race in Massachusetts, you need to raise a minimum of $2.5 million....
As you read this, you are probably wondering why the heck I am writing an editorial on complex aortic aneurysms when you know I...
FastWave Medical has announced the 30-day results of its first-in-human (FIH) study using the company's differentiated peripheral intravascular lithotripsy (IVL) technology.
This was a...
This month, I wanted to touch on that ubiquitous topic of failure. This often takes the form of not passing tests, or not matching,...
The Society for Vascular Surgery (SVS) has unveiled its strategic plans for fiscal year 2024–2025. On April 2, the SVS Executive Board (EB) and...
The 2024 Association of Program Directors in Vascular Surgery (APDVS) convened in Chicago April 5–6 for their annual spring meeting, which held the record...
As you read this, you are probably wondering why the heck I am writing an editorial on complex aortic aneurysms when you know I...
The SVS Foundation has announced recipients for the 2023–2024 awards cycle. The slew of awardees is headed by the winner of the S.T. Yao...
Karith Foster, chief executive of Inversity Solutions, is set to headline the inaugural Society for Vascular Surgery (SVS) Keynote Speaker Series at the 2024...
Imagine a world in which the enduring Dacron graft, birthed by vascular pioneer Michael DeBakey, MD, some 70 years ago as an open repair...
New data from the C-GUARDIANS pivotal investigational device exemption (IDE) trial support consideration of carotid artery stenting (CAS) with the CGuard embolic prevention stent...
New multisociety clinical practice guidelines for the management of lower extremity peripheral arterial disease (PAD) have been published online in the Journal of the...
SVS secretary details the spirit and the cause behind the next phase of the Society’s branding campaign aimed at helping vascular surgery become an...
New clinical results highlight the need for inclusive approaches and comprehensive examinations of treatment options for peripheral arterial disease (PAD), including endovascular therapy and...
This advertorial is sponsored by Penumbra, Inc.
“Honestly, there’s nothing even close to it for thrombectomy on the arterial side of things,” according to Martyn...
The inaugural president spoke to Vascular Specialist about the newly formed International Society for Women Vascular Surgeons ahead of this year’s Women’s Vascular...
In this issue:
Questions linger: BASIL-3 does not find for drug-eluting technologies for CLTI
Guest Editorial: Aortas, open surgical skills and gratitude
Vascular training:...
This advertorial is sponsored by Thompson Retractor.
Aortic aneurysm procedures range from basic to extremely complex. They all have one thing in common: great exposure...
Leading vascular surgeons from across the globe contributed pearls of wisdom on how to go about tackling inevitable turf battles from other specialties practicing...
In April, the most read stories from Vascular Specialist include new research on Chat generative pre-trained transformer (GPT) technology and its application within vascular...
Gore has announced US Food and Drug Administration (FDA) approval of an expanded indication for the Gore Excluder conformable abdominal aortic aneurysm (AAA) endoprosthesis....
On the final day of the 2024 International Vein Congress (April 18–20) in Miami, Florida, Joseph P. Hart, MD, associate professor of surgery and...
Patients undergoing cyanoacrylate glue closure for superficial venous disease reported higher periprocedural satisfaction than those who were treated via surgical stripping, data from a...
The Centers for Medicare & Medicaid Services (CMS) are exploring strategies to enhance the Quality Payment Program (QPP), underscoring an effort to boost healthcare...
The SVS emphasizes the role vascular surgeons play in advancing treatments for vascular diseases, offering a range of benefits through its membership to support...
As this year's Society for Vascular Surgery (SVS) Vascular Annual Meeting (VAM) approaches, it marks the end of an era for Andres Schanzer, MD,...
The American Board of Surgery (ABS) has unveiled the 15 proposed core Entrustable Professional Activities (EPAs) that will be evaluated in vascular surgery residents.
They...
ChatGPT 4.0 was able to provide a mean accuracy rating of 4.4 on a Likert scale of 5 (1=very poor; 5=excellent) across 15 patient-level...
A statewide collaborative between 35 hospital systems and a large insurance provider showed that the implementation of a performance metric linked with financial reimbursement...
In places across the country, it's spring. March Madness is ending, flowers are sprouting, gardens are being planted, and trees are budding. In the...
Abbott has announced that the US Food and Drug Administration (FDA) has approved the Esprit BTK everolimus-eluting resorbable scaffold system (Esprit BTK system), a...
In a podium-first presentation on the final day of the 2024 Charing Cross (CX) International Symposium (23–25 April, London, UK), Sean Lyden (Cleveland Clinic,...
In the UK National Institute for Health and Care Research (NIHR) Health Technology Assessment (HTA)-funded BASIL-3 randomised controlled trial (RCT), neither drug-coated balloon (DCB)...
“The surgical challenges of repairing deep venous reflux have been present for more than a generation. It is worth reflecting on the fact that...
Penumbra has announced the US Food and Drug Administration (FDA) clearance and launch of Lightning Flash 2, the next generation computer assisted vacuum thrombectomy (CAVT)...
The World Federation of Vascular Societies (WFVS) is bringing the vascular world together at CX 2024 to chew over both challenging cases and leadership...
“Unfortunately, we are doing worse for our patients today,” were the sobering thoughts of Eric Secemsky (Boston, United States) during a late-breaking presentation in...
The Society for Vascular Surgery (SVS) Executive Board has established the Vascular Board Certification Model Task Force to deliver a final report entitled “Free-standing...
The Society for Vascular Surgery (SVS) has announced Linda Harris, MD, and Palma Shaw, MD, as the candidates for the upcoming election for SVS...
As the Society for Vascular Surgery (SVS) prepares to launch its fifth cohort of the Leadership Development Program (LDP), anticipation is high among aspiring...
The progress in congress can seem overwhelmingly slow, especially considering the 118th Congress has only passed 27 pieces of legislation while holding over 700...
New research on Chat generative pre-trained transformer (GPT) technology and its Vascular Education and Self-Assessment Program (VESAP) success rate provides insight into the future...
Researchers behind a seven-year retrospective analysis of a physician-developed unitary stent graft system for endovascular debranched aortic repair of various thoracoabdominal aortopathies reported a...
Dawn M. Coleman, MD, chief of vascular surgery at the Duke University School of Medicine in Durham, North Carolina, focused on the likely major...
Five frequently performed vascular surgeries at a prominent New Orleans-based health system between 2019 and 2022 yielded an average yearly decrease in net revenue...
Increasing functional status in vascular patients fitted with a prosthesis after amputation was incrementally associated with improved survival, a retrospective analysis of a 10-year...
Though the 2024 edition of the Charing Cross (CX) International Symposium (April 23–25) in London, England, marks a new era, it will be tinged...
There has been much talk recently on open access publishing and the costs associated with this mode of publication. While editor-in-chief positions within most...
In this issue:
Bad reading: Worrying new data demonstrate a consistent shortfall of more than a fifth in net revenues over a four-year period...
In March, the most read stories from Vascular Specialist include a new analysis which suggests of a ‘lack of reach’ of vascular voices within...
SCVS 2024 saw Zachary AbuRahma, DO, an assistant professor of vascular surgery at West Virginia University and the Charleston Area Medical Center in Charleston,...
Evaluation of the safety and performance of a novel pharmaco-mechanical procedure for acute deep vein thrombosis (DVT) in two pigs demonstrated a significant reduction...
Johnson & Johnson is to acquire Shockwave Medical, it has been announced today.
Under the terms of the transaction, Johnson & Johnson will acquire all...
During the Society of Interventional Radiology (SIR) 2024 annual meeting (23–28 March, Salt Lake City, USA), newly presented data from a subgroup analysis of...
Registration for the 2024 Vascular Annual Meeting (VAM 2024) launched on March 20 and promotions teased a new annual VAM fixture: the SVS Keynote...
During a presentation at the 2024 European Vascular Course (EVC; March 3–5) in Maastricht, The Netherlands, Anna Pouncey, MD, a clinical research fellow for...
Getinge and Cook Medical today announced an exclusive sales and distribution agreement for the iCast covered stent system, which recently received Food and Drug...
Twelve-month outcomes from the Pounce venous thrombectomy system (Vetex Medical) first-in-human study evaluating its use in the treatment of acute iliofemoral deep vein thrombosis...
At the Charing Cross (CX) Symposium 2024 (23–25 April, London, UK), Andrew Bradbury (University of Birmingham, Birmingham, UK) and the BASIL-3 team of triallists...
Elixir Medical has announced Breakthrough Device designation by the Food and Drug Administration (FDA) for its novel DynamX BTK System, an implant for use...
Predicting longevity in patients who may be candidates for aortic repair is challenging. This, coupled with a high and unchanging long-term postoperative mortality rate,...
Cook Medical and Bentley today announced a distribution agreement for the BeBack crossing catheter in the U.S., with Cook assuming commercial responsibilities for the...
Jean Bismuth, MD, took over as Society for Clinical Vascular Surgery (SCVS) president during the organization's 51st Annual Symposium in Scottsdale, Arizona (March 16–20).
The...
Hans-Henning Eckstein, the vascular surgeon who played a leading role in the SPACE and SPACE 2 randomised controlled trials on the treatment of carotid...
Aquedeon Medical has announced the initiation of an investigational device exemption (IDE) clinical trial to study the Duett vascular graft system.
A press release reveals...
This advertorial is sponsored by Tampa General Hospital.
As Dean Arnaoutakis, MD, describes it, failed endovascular aortic aneurysm repair (EVAR) for abdominal aortic aneurysms (AAAs)...
Ruth Bush, MD, associate dean in the Office of Educational Affairs at John Sealy School of Medicine, the University of Texas Medical Branch (UTMB),...
Ticket information is available for the SVS Foundation’s “Night at the Museum—A Celebration of Science” Gala during the 2024 Vascular Annual Meeting (VAM 2024)...
The Society for Vascular Surgery (SVS) is encouraging members to pre-purchase the sixth edition of the Vascular Education and Self-Assessment Program (VESAP6), a premier...
The SVS has opened the nomination period for the position of SVS vice president, with the deadline set for March 15. Anyone can nominate/self-nominate...
In 2023, the SVS collaborated with Phairify to launch a compensation study to provide vascular surgeons with tailored insights into compensation, practice and productivity...
Society for Vascular Surgery (SVS) experts are spearheading initiatives as part of the multispecialty PAD Pulse Alliance and its Get a Pulse on PAD...
Filip Swirski, PhD, a leading figure in cardiovascular immunology research, will deliver this year’s Alexander W. Clowes Distinguished Lecture at the 2024 Vascular Research...
Data from a new study that maps out the levels at which adverse postoperative events decrease following transfemoral carotid artery stenting (TfCAS) based on...
Biotronik has been granted Breakthrough Device designation (BDD) from the US Food and Drug Administration (FDA) for the Freesolve below-the-knee (BTK) resorbable magnesium scaffold...
Alik Farber, MD, professor of surgery and radiology at Boston University, is set to present the results of a multicenter U.S. study at the...
A nearly record-breaking 794 Abstracts were submitted for the SVS 2024 Vascular Annual Meeting (VAM 2024). The abstracts were scored by the VAM Program...
In celebration of Women’s History Month, the Society for Vascular Surgery (SVS) Foundation’s “Voices of Vascular” series is underscoring women’s contributions to the specialty....
In the U.S., the legislative process is complex. Among the most important in this process are congressional committees, which play a pivotal role in...
One of the biggest mental shifts I had to make during residency was my view on failure. I literally had to rebrand failure. As...
My household had a longstanding Christmas tradition. In the months leading up to the big day, my wife would craft a gift budget for...
During the 2024 American Venous Forum (AVF) annual meeting in Tampa, Florida (March 3–6), Kurt S. Schultz, MD, a general surgery resident at the...
Integrated vascular surgery resident Eric Smith, MD, tells Vascular Specialist how he handled the lead up to Match Day.
For medical students hoping to specialize...
A new study set to be aired at the 2024 Society for Clinical Vascular Surgery (SCVS) annual meeting (March 16–20) in Scottsdale, Arizona, sheds...
This advertorial is sponsored by Inari Medical.
Several years ago, when Nicolas Mouawad, MD, division chief of vascular surgery at McLaren Health in Bay City,...
In this issue:
Learning curve: New data underscore physician experience levels required to derive benefit from transfemoral carotid artery stenting
New analysis points to...
The use of intravascular ultrasound (IVUS) for the treatment of claudication doubled over a four-year period but there was “no clear relationship between IVUS...
Rates of post-thrombotic syndrome (PTS) among deep vein thrombosis (DVT) patients treated with the ClotTriever thrombectomy system (Inari Medical) who are logged in the...
A large, single-center analysis has found that, despite parity between the two procedures in terms of perioperative clinical outcomes, carotid endarterectomy (CEA) was associated...
This year’s International Stroke Conference (Feb. 7–9) in Phoenix, Arizona, hosted discussions on the current state of play in carotid stenosis treatment—and, most notably,...
In February, the most read stories from Vascular Specialist include a study of metformin and its potential as the first-ever medical treatment that can...
At the 2024 American Venous Forum (AVF) in Tampa, Florida (March 3–6), Glenn Jacobowitz, MD, professor of vascular and endovascular surgery at NYU Langone...
A new study presented during the 2024 American Venous Forum (AVF) in Tampa, Florida (March 3–6), that compared a novel non-pneumatic compression device with...
An abstract presented at the 2024 American Venous Forum (AVF) annual meeting (March 3–6) revealed that, in the treatment of patients with pulmonary embolism...
Outgoing Vascular & Endovascular Surgery Society (VESS) President Mark Conrad, MD, used the 2024 VESS presidential address to illustrate how he got into vascular...
ViTAA Medical Solutions has announced the appointment of Frank J Veith, MD, professor of surgery at the Cleveland Clinic Lerner College of Medicine of...
Endologix has announced the initiation of the Percutaneous transmural arterial bypass (PTAB)1 postmarket study. This study marks the beginning of a comprehensive postmarket study...
New guidelines from the European Association for Cardio-Thoracic Surgery (EACTS) and the Society of Thoracic Surgeons (STS) have, for the first time, recognized the...
Andres Schanzer, MD, has hailed it "one of the most exciting changes" seen in imaging during the course of his career. Philips’ LumiGuide "human...
A nationwide U.S. study, recently published in the Journal of Vascular Surgery: Venous and Lymphatic Disorders (JVS-VL), showed that Black patients had the highest...
Mechanical thrombectomy using the ClotTriever device (Inari Medical) for iliofemoral deep vein thrombosis (DVT) was found to be “associated with significantly” lower Villalta scores...
The pitfalls of large administrative databases came to the fore during the presentation of a propensity-match scoring analysis demonstrating that percutaneous mechanical thrombectomy (MT)...
In a retrospective review of over 600 patients who underwent iliac vein stent placement, the development of back pain was found to be unrelated...
The European Union Horizon Europe, with joint funding from UK Research Innovation, has awarded the NetZeroAICT Consortium €6 million ($6.4 million) in funding to...
Pricing information for the 2024 Vascular Annual Meeting (VAM 2024) is now available. The event, scheduled in Chicago from June 19–22, is promoted as...
The SVS Executive Board has appointed the editorial team for the upcoming 12th edition of Rutherford’s Vascular Surgery and Endovascular Therapy. The team now...
The Journal of Vascular Surgery (JVS) editors announced the open-access articles available in the flagship title’s March 2024 issue.
The relevant papers are: “Creating hemodialysis...
The Journal of Vascular Surgery Cases, Innovations and Techniques (JVS-CIT) has published its latest virtual special issue, which covers genetic arteriopathy. This is the...
In the fast-evolving landscape of medical education, podcasts have emerged as a powerful medium for disseminating knowledge and fostering community among professionals. Among these,...
So, what is budget neutrality and how does it affect physician payment? The Medicare Physician Fee Schedule (PFS) is a resource-based relative value scale...
Vascular resident Saranya Sundaram, MD, shares a story of finding cultural understanding and community.
“It’s Sundaram, S-U-N…” I’m interrupted by a nurse asking me if...
The 2024 Southern Association for Vascular Surgery (SAVS) annual meeting (Jan. 24–27) hears three separate talks—two scientific papers and one from an invited guest...
The American College of Surgeons (ACS) and the Society for Vascular Surgery (SVS) have verified the first three outpatient centers under the newly launched...
The American Heart Association (AHA) has named the BEST-CLI (Best endovascular vs. best surgical therapy in patients with critical limb ischemia) trial one of...
A randomized controlled trial (RCT) of open repair versus endovascular aneurysm repair (EVAR) for the treatment of abdominal aortic aneurysms (AAAs), as well as...
The fifth cohort in the society for Vascular Surgery (SVS) Leadership Development Program (LDP) reached capacity in early February due to limited space and...
In response to recent articles in the lay press suggesting vascular surgeons were performing unnecessary procedures, Vascular Specialist medical editor, Malachi Sheahan, has asked...
Sensome has announced enrollment of the first patients into a feasibility clinical study using the Clotild smart guidewire in peripheral arterial disease (PAD). Clotild was designated...
Amid the build-up to residency Match Day 2024, the Association of Program Directors in Vascular Surgery (APDVS) has delivered data from a VISIT (Vascular...
In this issue:
SAVS outgoing President David L. Cull details his journey to the heart of diversity, equity and inclusion
More than half of...
During Black History Month, the Society for Vascular Surgery (SVS) Foundation will shine a spotlight on the contributions of Black vascular surgeons through its...
The SVS will host a webinar Feb. 29 to help equip members with insights on impending Centers for Medicare & Medicaid Services (CMS) reporting...
An upcoming webinar is set to provide insights for healthcare professionals aiming to enhance their expertise in treating patients with chronic limb-threatening ischemia (CLTI)....
Efemoral Medical has announced the granting of Food and Drug Administration (FDA) Breakthrough Device status for its novel Efemoral vascular scaffold system (EVSS) designed...
David L. Cull, MD, delivers a captivating presidential address, laying bare his own challenging journey as a vascular surgeon and the hidden lessons on...
In January, the most read stories from Vascular Specialist include a U.S. Aortic Research Consortium analysis of F/BEVAR preoperative risk factors in AAAs/TAAAs; a...
The uncommonly performed aortoiliac endarterectomy—one of the open procedures on which the vascular surgical specialty was founded—provides acceptable durability in aortoiliac occlusive disease patients...
The Society for Vascular Surgery (SVS), in partnership with three other medical societies operating in the vascular disease space that form the PAD Pulse...
The use of a relatively novel scoring system, referred to as Symptomatic carotid atheroma inflammation lumen-stenosis (SCAIL), has produced favourable predictive qualities versus the...
Following a review, the United Kingdom's Medicines and Healthcare Products Regulatory Agency (MHRA) has updated guidance on the use of paclitaxel-coated devices stating that such devices...
A study comparing the extent of early infrapopliteal recoil after serration and plain balloon angioplasty has found the former produces “substantially less” arterial recoil...
The SVS foundation celebrated another philanthropic success through its Giving Tuesday efforts, raising $20,000 in funds. Presenting sponsor W. L. Gore and Associates doubled...
In the late 1970s, a neurosurgeon at New York’s St. Luke’s Hospital operated on a beautiful, young, rising soprano who studied opera at a...
The SVS is set to launch the sixth edition of the Vascular Education and Self-Assessment Program (VESAP6) in April. The highly anticipated review resource...
The Society for Vascular Surgery (SVS) reflects on a year marked by notable achievements on multiple fronts. In a communication to members in mid-December,...
The first patient has been enrolled into the HERCULES trial, which compares endosuture aneurysm repair (ESAR) to standard endovascular aneurysm repair (EVAR) in the treatment of abdominal aortic...
There is a global interest in assessing whether metformin, which has a long track record of safety and efficacy, is relatively inexpensive and is...
My limited understanding of psychological safety is when people feel safe expressing differing opinions, and thoughts in the workplace. Amy Edmondson, a pioneer in...
Cook Medical has announced the first patient treated in the clinical study of the Zenith Fenestrated+ endovascular graft (ZFEN+) in the U.S. The procedure was...
The SVS is accepting applications for vice chairs and volunteers. SVS members in good standing can apply for up to two committee positions and...
The SVS has expanded its Mentor Match program, which is now available to all SVS members in good standing. The program aims to connect...
The SVS Sub-Section on Outpatient and Office Vascular Care (SOOVC) has released the anticipated Office-Based Lab (OBL) Handbook to guide practitioners and aid with...
Gore has announced recent Food and Drug Administration (FDA) approval of a lower profile Viabahn VBX balloon expandable endoprosthesis (VBX stent graft) for the treatment...
Endospan shared 30-day results from the first 22 patients enrolled in the TRIOMPHE investigational device exemption (IDE) study in a late-breaking presentation at the...
The Southern Association for Vascular Surgery (SAVS) elected Margaret (Megan) Tracci, MD, to become its first ever woman president for the 2025–26 cycle.
Tracci,...
The new year is a time for reflection. It is also an opportunity to rededicate, or if needed, reinvent ourselves. Vascular surgery training is...
Gore has announced Food and Drug Administration (FDA) approval for the Gore Excluder thoracoabdominal branch endoprosthesis (TAMBE), which the company states is the first...
The updated European Society for Vascular Surgery (ESVS) 2024 clinical practice guidelines for the care of patients with aneurysms of the abdominal aorta and...
A vascular surgery team at Audie L. Murphy VA Medical Center in San Antonio, Texas, uncovers “staggeringly low” numbers of correctly coded billing for...
The final publication of midterm results from the observational CARAS study suggest that cerebral ischaemic event (CIE) incidence in asymptomatic carotid stenosis patients “should...
Last October, the University Pittsburgh’s Clinical and Translational Sciences Institute hosted its 10th Pitt Innovation Challenge (PinCh), providing a total of $550,000 in prizes,...
The International Working Group on the Diabetic Foot (IWGDF), the European Society for Vascular Surgery (ESVS) and the Society for Vascular Surgery (SVS) have...
FastWave Medical has announced the successful completion of enrollment for its first-in-human (FIH) study with the company’s peripheral intravascular lithotripsy (IVL) technology.
The prospective, single-arm...
This advertorial is sponsored by Tampa General Hospital.
Early on, Jean Bismuth, MD, acquired a passion for vascular surgery from a world-class source. Back in...
Gore has announced that the first patients have been enrolled in the Gore VBX FORWARD clinical study, a global prospective, multicenter, randomized controlled trial to compare...
In this issue:
Coding: Increasing complexity and lost RVUs—a drop in the ocean?
The utility of renal stenting in hemodialysis patients: one in five...
A series of preoperative risk factors—including currently smoking, chronic kidney disease (CKD), congestive heart failure (CHF), aneurysm size greater than 7cm, more advanced...
A fifth of patients among a chronic kidney disease (CKD) cohort on hemodialysis—a rare subset pulled from a large repository of national data—were able...
A dedicated session at the 2023 VEITHsymposium (Nov. 14–18) in New York City aimed to unpack the ways in which clinical practice and attitudes...
Carotid artery stenting (CAS) delivered via transradial artery access has demonstrated no increased risk of stroke, death, myocardial infarction, transient ischaemic attack, or access...
Endovascular Engineering (E2) has announced US Food and Drug Administration (FDA) investigational device exemption (IDE) approval for its ENGULF US pivotal trial.
The study will...
Proceedings from an expert consensus roundtable that discussed the benefits of intravascular ultrasound (IVUS) in lower extremity revascularization procedures were released today in the...
The Society for Vascular Surgery (SVS) is an active participant in the American Medical Association (AMA)/ Specialty Society Relative Value Scale Update Committee (RUC)...
Endologix recently announced that the Centers for Medicare & Medicaid Services (CMS) has granted a Transitional Pass-Through (TPT) payment for the Detour system, effective...
The Society for Vascular Surgery (SVS) Resident and Student Outreach Committee (RSOC) is accepting applications through Jan. 8, 2024, for the Diversity Medical Student...
Which stories captured the attention of the vascular community over the course of the past year? Read our summary of the trending articles from...
While advocacy consistently maintains a top spot among the list of priorities for SVS members, there remains a degree of uncertainty among many in...
Christopher Audu, MD, surveys the 2024 meeting landscape, highlighting prime opportunities for trainees to showcase their research.
It is abstract submission, and this month I...
In a tribute to the late James S.T. Yao, MD, PhD, a luminary in the field of vascular surgery, the Yao family and the...
The Society for Vascular Surgery (SVS) is calling for nominations for its Excellence in Community Practice Award, which recognizes outstanding leadership within the community...
Gore has announced the first patient implantation of the company's ascending stent graft in the ARISE II trial, describing this as an exciting step...
“We’re going to see more and more amputations if we don’t figure out the right post procedure thromboprophylaxis regime ASAP,” Anahita Dua, MD, warned...
The Journal of Vascular Surgery: Venous and Lymphatic Disorders (JVS-VL) will move from a subscription model to one of open access—freely available online—on Jan....
SVS members are taking advantage of the new Smoking Cessation Toolkit and resources from the Vascular Quality Initiative (VQI), adding them to existing programs...
Proper integration of primary care physicians (PCPs) in the multidisciplinary care of vascular wounds is going to play an increasingly important role as the...
The Society for Vascular Surgery (SVS) has issued a statement addressing the latest reports published by ProPublica on atherectomy use in the U.S.
The nonprofit...
Christopher J. White, MD, responds to a recent editorial by Malachi Sheahan III, MD, that took a critical look at the decision by the...
The Society for Vascular Surgery (SVS) has received a $100,000 grant from the Council of Medical Specialty Societies (CMSS) to promote diagnostic excellence across...
Humacyte today announced that it has submitted a Biologics License Application (BLA) to the Food and Drug Administration (FDA) seeking approval of the Human...
Our biases permeate the fabric of our very being, as they weave their way into our training and practice. It is clear that complex...
The recent move to expand Medicare coverage of carotid stenting formed the basis of a recent debate over the wisdom of the decision taken...
The emergence of a baroreflex activation therapy device for heart failure patients is drawing in the assistance of the open surgical expertise of vascular...
The voting members of the SVS have approved amendments to the Society bylaws following a referendum that closed on Nov. 20. The proposed changes...
The Society for Vascular Surgery (SVS) has made a fresh call for members to participate in a compensation survey, a collaboration with Phairify, that...
In the end, reflected Michael S. Conte, MD, “it’s not the evidence.” The leading vascular surgeon and peripheral arterial disease (PAD) expert made the...
Patient enrollment is now complete in the APEX-AV trial evaluating the safety and efficacy of the AlphaVac F1885 (AngioDynamics) multipurpose mechanical aspiration system for...
In this issue:
Changing course: The natural history of tibial claudication comes under scrutiny as interventions for "relatively benign" disease trend upward
Our guest editorial...
In November, the most read stories from Vascular Specialist include a reassessment of which carotid revascularization treatment modality is best after the recent Centers...
A new intravenous anesthesia delivery technique used during endothermal ablation for varicose veins demonstrated comparatively low pain scores according to first-in-human (FIH) data recently...
New analyses from the VOYAGER PAD clinical trial in both high-risk and fragile patients and those with and without comorbid coronary artery diseases (CAD)...
Researchers may now submit abstracts for the SVS Vascular Research Initiatives Conference (VRIC), to be held in conjunction with the American Heart Association’s...
SVS members are reminded to act promptly before the year’s end to ensure uninterrupted access to their 2024 membership benefits.
Renewals are due on...
Peter Schneider (University of California San Francisco, San Francisco, USA) recently presented 36-month data from Surmodics' TRANSCEND clinical trial at the VEITHsymposium 2023 (14–18...
Now set to welcome its fifth cohort, the SVS Leadership Development Program (LDP) continues to help vascular surgeons across the U.S. and Canada to...
As the holiday season comes into focus, the SVS Foundation is preparing to celebrate science and honor our vascular heroes on Giving Tuesday, a...
The SVS Sub-Section on Outpatient and Office Vascular Care (SOOVC) has opened applications for its Presentation Award and Research Seed Grant, allowing practitioners to...
VAM program chair Andres Schanzer, MD, makes a call for cutting-edge new science for next year’s meeting in Chicago.
Researchers who want to present new...
“We have a critical problem in America, which is the under-appreciation of peripheral arterial disease ,” says Vikram Kashyap, MD, on his first time...
Change is a constant in life, and advocacy is no exception. The healthcare landscape is always evolving, and the SVS Advocacy Council is working...
Nicolas Mouawad, MD, chief of vascular surgery at McLaren Health System in Bay City, Michigan, urges vascular surgeons to “get out of their comfort...
Xeltis has announced "highly encouraging" 12-month results from its first-in-human (FIH) aXess vascular conduit trial. The data were presented by Frans Moll (University Medical...
An ongoing UK National Institute for Health and Care Research (NIHR)-funded randomized controlled trial seeks to illuminate the value of an image fusion guidance...
This summer the center for medicare and Medicaid Services (CMS) published its annual Physician Fee Schedule and Proposed Rule for calendar year 2024, and...
Two-year data from the SWING trial, a first-in-human study of the safety and performance of the Sundance sirolimus drug-coated balloon (DCB; Surmodics), have been...
I enjoy listening to podcasts. In addition to my usual rotation through Audible Bleeding and Behind the Knife, I’ve found insight in listening to...
Periodically, when under pressure at work, and sometimes at home, I would think about what I could do to free up more time for...
A 14-year effort to achieve departmental status for vascular surgery at a large healthcare system in Southern California resulted in significant gains for the...
Vascular surgeon-scientist Elsie Gyang Ross, MD, recently secured coveted funding through the National Institutes of Health (NIH) Research Project Grant Program (R01)—a rare feat...
The Society for Vascular Surgery (SVS) Executive Director Kenneth M. Slaw, PhD, has been elected to executive council of the Council of Medical Specialty...
In this issue:
Carotid revascularization: Getting to the right decision
SVS reveals proposed changes to Society bylaws
Meta-analysis: The complete patient-level dataset on paclitaxel and...
This advertorial is sponsored by Silk Road Medical.
For Scott Berman, MD, patient responses when confronted with an explanation of the extent of their asymptomatic...
This advertorial is sponsored by AOTI.
Enrico Ascher, MD, and Natalie Marks, MD, currently combine for close to 60 years of vascular surgery practice experience...
The recent move by the Centers for Medicare & Medicaid Services (CMS) to expand coverage for carotid artery stenting brought with it the requirement...
Endologix has announced results from a pooled analysis of the DETOUR1 and DETOUR2 studies evaluating percutaneous transmural arterial bypass (PTAB) with the Detour system.
The Detour system...
This advertorial is sponsored by Philips.
The benefits of utilizing intravascular ultrasound (IVUS) in peripheral vascular interventions has been widely demonstrated in the literature. In...
One-year results from the PROMISE II US pivotal trial demonstrate the durability of outcomes of transcatheter arterialisation of the deep veins (TADV) using the...
Last month, the most popular stories from Vascular Specialist included The Centers for Medicare & Medicaid Services (CMS) confirmation of carotid artery stenting coverage expansion;...
Thirty-day results from two trials assessing the performance of investigational carotid artery stenting systems—the Neuroguard and the CGuard—were recently presented at VIVA 2023.
Stenting with...
LimFlow announced today that it has entered into a definitive agreement to be acquired by Inari Medical.
A press release details that, under the terms...
Patients undergoing computer-assisted vacuum thrombectomy with the Indigo Aspiration System (Penumbra) for pulmonary embolism (PE) showed 2.7% rates of both major adverse events (MAEs)...
The SVS has announced an expansion of its coding-related information and help in response to increasing member demand. The new resources will assist vascular...
A recently published study has shown that lower limb bypass surgery offered a significantly higher probability of amputation-free survival and wound healing compared with...
November is National Diabetes Month. Because SVS members treat patients with the condition—and its complications—the Society has numerous educational resources available.
“Diabetes and Vascular Disease”...
Results from a first-in-human, prospective, single-arm study of the Akura Medical thrombectomy system (Akura Medical) for pulmonary embolism (PE) were revealed this week at...
A secondary analysis of existing clinical trial data has indicated that new ischaemic brain lesions detected via diffusion-weighted magnetic resonance imaging (DW-MRI) following carotid...
The open-label phase of the DEXTERITY-AFP trial investigating the Bullfrog microinfusion device—which involves the perivenous injection of the anti-inflammatory drug dexamethasone to improve patency...
David Dexter (Sentara Vascular Specialists, Norfolk, USA) shared one-year data from the CLOUT registry this week at The VEINS 2023 (28–30 October, Las Vegas,...
The SVS, in collaboration with the American Venous Forum (AVF) and the American Vein and Lymphatic Society (AVLS), has unveiled part two of...
In September, the Society for Vascular Surgery submitted a detailed comment letter to the Centers for Medicare and Medicaid Services (CMS) in response to...
Data can be granular and clarifying. Conversely, data without appropriate context can lack nuance—like a paint-by-numbers portrait using only one crayon.
A recent article publivenshed...
With the congressional “busy season” upon us, the SVS is gearing up for big tasks ahead on the legislative and regulatory fronts. Having stepped...
Voting members of the SVS will have the opportunity to consider a bylaws referendum, with key amendments related to membership and the composition of...
SVS members must act before the end of the year to avoid interruption of their 2024 membership benefits. Members can pay their dues on...
Applications open this month for the SVS Excellence in Community Practice Award and the Sub-Section on Outpatient & Office Vascular Care (SOOVC) Research Seed...
Data from the REAL-PE study were presented this week at TCT 2023 (23–26 October, San Francisco, USA) demonstrating that patients treated for pulmonary embolism...
Thrombolex has announced never-before-reported major reductions in obstruction in all of the segmental pulmonary arteries (PA), based on independent core lab data analysis of...
During the vascular annual meeting that celebrates science and research, the SVS Foundation’s annual Gala will take place in a museum that also honors...
This month in Corner Stitch, we highlight a trainee-centric study presented at the 2023 Midwestern Vascular Surgical Society (MVSS) annual meeting held in Minneapolis,...
The September issue of Annals of Vascular Surgery featured 37 original, peer-reviewed scientific papers whose lead authors and researchers are women vascular surgeons. Here,...
Julie A. Freischlag, MD, used her turn as honorary guest lecturer at the 2023 Midwestern Vascular Surgical Society (MVSS) annual meeting to make a...
Results of the LIFE-BTK randomized controlled trial have just been presented at TCT 2023 (Oct. 23–26) in San Francisco. The data show that, in...
Data from a patient-level meta-analysis—a factor in the Food and Drug Administration’s (FDA) recent change of position on the use of paclitaxel-coated devices to...
Data from a patient-level meta-analysis—a factor in the US Food and Drug Administration’s (FDA) recent change of position on the use of paclitaxel-coated devices...
The majority of more than 300 vascular surgeons who responded to a recent survey indicated that they perceive the management of venous disease to...
Robert Cambria, MD, recently took over as president of the New England Society for Vascular Surgery (NESVS). He was passed the presidential baton by...
Organizing a Vascular Annual Meeting (VAM) takes a whole lot of time and a whole lot of long-range planning.
Thus, members of the SVS Postgraduate...
New research examining the safety and efficacy of using radial access for peripheral artery interventions has found that radial access allows early ambulation and...
An inferior vena cava (IVC) filter retrieval device dubbed the next-generation in removal of the venous thromboembolism-fighting tools could substantially cut procedural times and...
A vascular surgeon's value includes far more than direct revenues from procedures performed, said SVS member Robert Tahara, MD, recently. He told surgeons to...
The SVS Foundation's Board of Directors announced the appointment of two new board members, Edith Tzeng, MD, and Palma Shaw, MD, at the board’s...
This September, 533 people put their feet to the ground to raise awareness about peripheral arterial disease (PAD) as part of the SVS Foundation’s...
A mass shooting in Las Vegas. A bombing at the finish line of the Boston Marathon. A shooting at a Fourth of July parade....
The Centers for Medicare & Medicaid Services (CMS) has released its final decision regarding National Coverage Determination (NCD) 20.7 covering carotid artery stenting (CAS),...
While sex disparities regarding the outcomes of carotid revascularization have “long been a concern,” new prospective data published in the Journal of Vascular Surgery...
A virtual roundtable on Oct. 10 will cover the new Society for Vascular Surgery (SVS) varicose veins guidelines. Set for 6–7:30 p.m. CDT, the...
First, a disclaimer. This commentary is woefully short of sex-related content. Not even the wholesome gender-based outcomes stuff. So, if that key term brought...
Contego Medical has announced enrollment of the first patient in the prospective, multicenter PERFORMANCE III trial aimed at further evaluating the safety and effectiveness of the...
Roger Malcolm Greenhalgh, MD, the surgeon internationally renowned for his unparalleled contribution to vascular education, training and research, died peacefully on Oct. 6. He...
This advertorial is sponsored by Inari Medical.
For patients with in-stent thrombosis (IST), one of the most critical unmet needs has been an effective method...
In this issue:
Preventing spinal cord ischemia: Novel techniques for direct segmental artery revascularization show promise
Eastern Vascular: New data chart underrepresentation of women...
The underrepresentation of women vascular surgeons in national clinical trial leadership formed the central theme of data coming out of a new study that...
George S. Lavenson Jr., lovingly referred to as “Doc” by his many friends, died Saturday, Aug. 19, 2023, on the beautiful island of Maui,...
An investigator-sponsored clinical study conducted at the Mayo Clinic in Rochester, Minnesota, of the investigational Human Acellular Vessel (HAV) in patients with chronic limb-threatening...
Outgoing Midwestern Vascular Surgical Society (MVSS) President Jeffrey Jim, MD, used his recent presidential address to call upon vascular surgeons to spread the word...
On March 22, 1978, Karl Wallenda went up on a high wire in Puerto Rico strung between two high-rise buildings. There was no safety...
Last month, the most read stories from Vascular Specialist included data releases from the recent Midwestern Vascular Surgical Society (MVSS) using direct revascularization of segmental...
The potential for large language-learning models like ChatGPT to take over tedious writing tasks such as emails and parts of PowerPoint presentations—or even be...
Outgoing Western Vascular Society (WVS) President Wei Zhou, MD, used her presidential address to put out a call for the vascular specialty to embrace...
The Journal of Vascular Surgery: Venous and Lymphatic Disorders (JVS-VL) has launched its first virtual special issue on diversity, equity and inclusion (DEI) in...
The SVS History Digital Archives Task Force is soliciting contributions as it aims to present a curated collection to SVS members and the medical...
In the dynamic arena of healthcare advocacy, the Society for Vascular Surgery (SVS) stands as a champion, harnessing the power of amplification to thrust...
A vascular surgery team from the University of Southern California (USC) in Los Angeles demonstrated the feasibility of multidisciplinary direct revascularization of segmental arteries...
Laminate Medical Technologies has announced their flagship device, the VasQ external vascular support, has been cleared by the Food and Drug Administration (FDA) for...
Viz.ai has announced new clinical data supporting advancements in pulmonary embolism (PE) detection. Two studies have demonstrated the real-world clinical efficacy of Viz.ai’s PE module to...
“Voices of Vascular,” an initiative to highlight the diverse voices within the SVS, has earned the Society and the SVS Foundation a Profile of...
This commentary—authored by Anne Abbott, PhD, MBBS, Lawrence Schott, MD, Lan Gao, MMed, PhD, Hrvoje Budincevic, MD, PhD, Rishad Faruqi, MD, Tatjana Rundek, MD,...
Roy M. Fujitani, MD, president of the University of California, Irvine (UCI) Medical Center medical staff, took over as Western Vascular Society (WVS) president...
The Society for Vascular Surgery (SVS) and the American College of Surgeons (ACS) have released new standards for outpatient vascular centers to help them...
The Southern Association for Vascular Surgery (SAVS) has put out a call for applications from medical students and general surgery residents for 20 travel...
The 2024 Society for for Vascular Surgery (SVS) Vascular Research Initiatives Conference (VRIC), which concentrates on basic and translational vascular science, will take place...
“We face a scarcity of data evaluating endovascular therapy for isolated popliteal artery lesions, known as a difficult vessel bed to treat as we...
“Multifactorial” issues persist in gender representation in vascular leadership and trial enrollment, leading women vascular surgeons tell Vascular Specialist.
"We would have fewer untoward side...
Humacyte has announced positive top line results from its V005 Phase 2/3 trial of the Human Acellular Vessel (HAV) in vascular trauma repair.
A...
The Society for Vascular Surgery’s Patient Safety Organization (SVS PSO) has formally launched a national smoking cessation initiative. It includes tools and resources to help physicians,...
The Food and Drug Administration (FDA) today announced approval of LimFlow therapy—a novel and minimally invasive procedure designed to bypass blockages in arteries of...
Bernadette Aulivola, MD, the division director of vascular surgery and endovascular therapy at Loyola University in Maywood, Illinois, took over as president of the...
Kathleen Ozsvath, MD, a vascular surgeon at St. Peters Health Partners in Albany, New York, took over as president of the Eastern Vascular Society...
Shape Memory Medical recently announced that the Food and Drug Administration (FDA) has granted investigational device exemption (IDE) for the company to begin a...
The Society for Vascular Surgery (SVS), American Venous Forum (AVF), and American Vein and Lymphatic Society (AVLS) have released the second and final part...
In a world in which both medical breakthroughs and technological advancements seem to dominate the healthcare landscape, it’s easy to overlook the unsung heroes...
Organizers behind an SVS-commissioned financial compensation survey are calling on more members to fill out the short list of questions so that they can...
In this issue:
When women are PIs: A story foretold on the vascular frontlines
SVS members rally to respond to CMS carotid stenting coverage...
The Centers for Medicare & Medicaid Services (CMS) in mid-July released two payment-related proposed rules—the highly-anticipated calendar year (CY) 2024 Medicare Physician Fee Schedule...
Organizers of VAM 2024 want members to consider what the modern-day vascular surgeon needs to know for optimal patient care and professional success. What’s...
Nearly one-third of the public comments submitted to the Centers for Medicare & Medicaid Services (CMS) on a controversial proposed coverage decision affecting carotid...
Welcome back to Government Grand Rounds, a campaign aimed at expanding vascular surgeons’ understanding of—and comfort with—the SVS’ many advocacy tools. In the dynamic...
Limited spots remain open for two popular SVS courses coming in October: the Complex Peripheral Vascular Interventions (CPVI) Skills Course on Oct. 1–2 and...
The month of September begins Friday and with it, the SVS Foundation’s Vascular Health Step Challenge.
The initiative challenges participants to walk 60 miles—representing the...
The first U.S. patient has been treated as part of the SOCRATES trial (Short neck AAA randomized trial—ESAR and FEVAR), which compares the safety...
In the wake of the coverage of inappropriate vascular interventions in the mainstream press, guest editorialist Adam Tanious, MD, tackles the thorny issue of...
NOTE: This video is ONLY available to watch in selected countries and geographies
The benefits of Shockwave Medical’s new Shockwave L6 Peripheral Intravascular Lithotripsy (IVL) catheter—designed...
Nectero Medical announced that the Food and Drug Administration (FDA) has granted Fast Track designation for the Nectero Endovascular Aneurysm Stabilization Treatment (Nectero EAST)...
The question of “persistent problems” with venous stenting among post-thrombotic syndrome (PTS) patients emerged as a key talking point during the recent Deep Venous...
There is no doubt that the Medicare payment system needs reform. At this moment, the SVS is aiming to inspire reform through all facets...
Cydar Medical has announced the successful treatment of the first patient in a strategic collaboration with Medtronic. The collaboration, which spans 40 sites across...
NOTE: This video is ONLY available to watch in selected countries and geographies
Experts gathered at the Society for Vascular Surgery’s 2023 Vascular Annual Meeting...
Clinical outcomes of Humacyte's investigational Human Acellular Vessel (HAV) were presented at the Military Health System Research Symposium in Kissimmee, Florida (Aug. 14–17), following...
In a recently published study, dedicated venous stents performed well through pregnancy and postpartum, and a protocol including the use of low-dose antiplatelets in...
Researchers report that inferior vena cava (IVC) filter placement position relative to the level of the most inferior renal vein was not associated with...
The SVS will host the SVS Coding and Reimbursement Workshop this fall, providing a comprehensive program designed to equip vascular surgeons and their support...
Phase II of a national branding campaign for vascular surgery and three new task forces are just two of several initiatives the SVS’ Executive...
Time is running out to plan attendance at the annual course, A Comprehensive Review and Update of What’s New in Vascular and Endovascular Surgery,...
As a vascular surgery trainee, there are multiple aspects of vascular surgery that I’ve had the opportunity to learn about. And to be honest,...
Because high-quality evidence from large, prospective multicenter trials on the best treatment of uncomplicated type B aortic dissection (uTBAD) is scarce, SVS members Firas...