
This advertorial is sponsored by Inari Medical.
For patients with in-stent thrombosis (IST), one of the most critical unmet needs has been an effective method to remove acute-to-chronic material. Here, Rishi Roy, MD, a vascular surgeon at Advanced Vascular & Vein Associates in Flowood, Mississippi, discusses his experience with the first mechanical thrombectomy device to be able to do so—the RevCore thrombectomy system.
RevCore is a minimally invasive, over-the-wire device indicated to treat thromboemboli in the peripheral vasculature, including venous stents. It includes a catheter with an expandable element for venous stent treatment. It also has a reinforced catheter shaft for precise movements and a diameter-control knob for adjusting the element to treat vessels 6mm-plus and venous stents from 10–20mm.
Prior to RevCore, how did you treat in-stent thrombosis?
Previously I would take a multimodal approach using suction thrombectomy, along with balloon venoplasty, or even a non-compliant balloon. The goal was to get some lumen gain, to restore some outflow to decompress the limb.
How does this new device work?
RevCore makes it possible to extract thrombotic material from an occluded stent. The diameter of the coring element can be increased or decreased manually to accommodate the size of the vessel . You engage or disengage with the stent, as needed, and you can be more aggressive as you work through the material that caused the occlusion.
How do you size the catheter and capture liberated clot?
When sizing the element in the stent, low and slow is key. There is a tactile feel to the device when “revving,” and it’s important to note resistance level. Some resistance is good—too much means the element is likely oversized and should be decreased by turning the external diameter-control knob to the minus sign on the handle. An important consideration for these cases is to have another device placed in the inferior vena cava (IVC) to capture the cleared material.
When do you know that RevCore worked?
Immediately postprocedure, I can gauge whether the device worked when I visualize blood flow on imaging. Because the device is new, I have an intensive follow-up protocol and, working with a wound care group, even closer follow-up is possible. I generally see the patient back in four weeks. After that, we see them at three and six months, and then every six months. If over a two-year period we don’t see recurrent stenosis, and the patient is comfortable, we could get it down to once a year.
What anticoagulation regimen do you give these patients?
I send them out on either a novel oral anticoagulant (NOAC) or Coumadin. You know, people often ask me, “Which do you prefer?” My honest answer is I prefer whatever the patient can afford. Because if they can’t afford a NOAC, then they’re not going to take it, and we’re back at square one.
How do you think RevCore will change the treatment pathway?
RevCore allows us to consider intervention sooner for patients who are suffering from symptoms related to IST. Some had stents placed when they were in their 20s or 30s, and when these stents are put in, they’re in forever. Now it’s decades later, and they have recurrent IST with chronic material that requires reintervention every three-six-eight months for symptom relief. Now, if we can debulk and get back to the original stent diameter, that patient will require close follow-up every six months or so, but they may be able to go for a couple of years between interventions. Fewer interventions also means decreased anesthesia time, operative time, chance of adverse events and morbidity. If we can decrease the number of cases because we’re able to remove IST in a single procedure, that’s a game changer.
Case report: Complete thrombus extraction after severe right EIV stent stenosis
Patient history
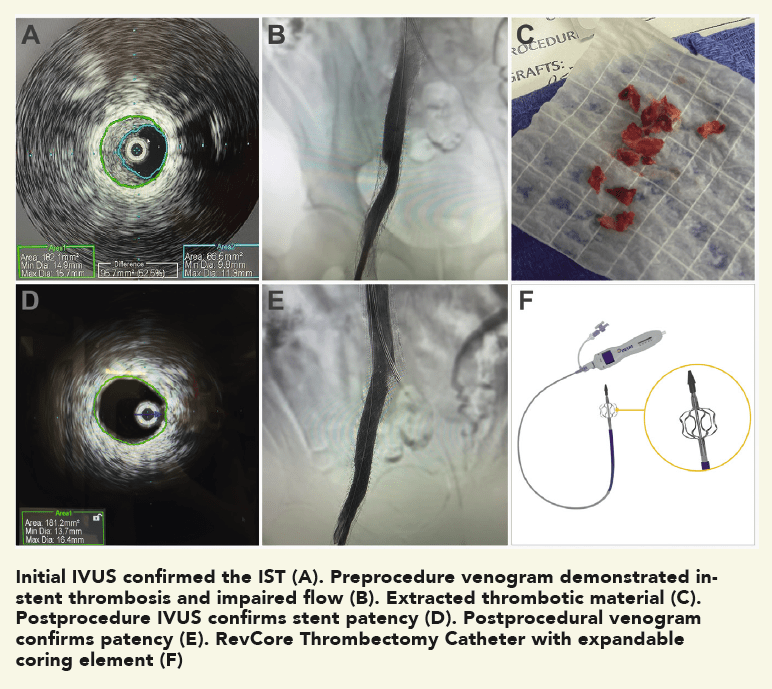
A man in his mid-50s presented with ulceration and swelling in his right foot with the intention to transfer care. Several months prior, a right external iliac vein (EIV) to right common femoral vein (CFV) stent had been placed. A venous duplex ultrasound study showed proximal stent occlusion and non-occlusive venous thrombosis in the left proximal femoral, popliteal, posterior tibial, and gastrocnemius vessels, as well as thrombosed varicosities. Right lower extremity venography and intravascular ultrasound (IVUS) performed two-and-a-half weeks later confirmed 65% stenosis of the left EIV stent (image A). An in-hospital mechanical thrombectomy and balloon angioplasty procedure was planned.
 Procedural overview
Procedural overview
The patient was positioned supine and ultrasound guidance was used to access the right CFV. A J-wire was advanced into the IVC. An 8F sheath was placed and a venogram showed stenosis within the right EIV stent (B). Similarly, access was gained to the left CFV, and a J-wire was advanced into the IVC. An 11F sheath was placed and a venogram confirmed there was no stenosis in the selected segments.
A FlowTriever catheter (Inari Medical) was advanced over a stiff wire to the infrarenal segment of the IVC, and two of the catheter’s XL disks were deployed. From the right side, a stiff wire was advanced to the IVC via a Bern catheter (Boston Scientific), followed by an Amplatz wire. The catheter was removed, and then a 16F sheath was placed.
IVUS was performed in the IVC, revealing that the stent extended into the very distal IVC; no stenosis was seen. IVUS of the right common iliac vein (CIV) showed overlapping stents but no stenosis. A venous ultrasound probe inserted to the level of the right EIV showed 50–60% stenosis. IV heparin was administered.
The RevCore catheter was advanced over the Amplatz wire to the right EIV, and the coring element was deployed within the stent in an unexpanded state. The coring element was then slowly enlarged and rotated, loosening the material within the stent. RevCore was then withdrawn and cleaned. A Triever16 Curve catheter (Inari Medical) was advanced and suction thrombectomy performed, removing a combination of subacute and chronic thrombotic material (C). Balloon venoplasty of the stented portion of the right CFV, EIV and CIV was performed with an 18mm noncompliant balloon.
Additional passes with the Triever16 Curve catheter were conducted along the right iliac segments and within the IVC, removing thrombotic material that had been captured by the FlowTriever XL disks. Thrombus capture with either FlowTriever XL disks or the Protrieve sheath is critical to the RevCore procedure. Blood was returned to the patient using FlowSaver.
A final IVUS showed nearly 100% lumen gain, restoring the right EIV to CFV in-stent diameter to 16.4 mm (D). A final venogram demonstrated appropriate flow through the right CFV, EIV, CIV and IVC, and resolution of the stenosis within the right EIV (E). The FlowTriever disks and all other devices were removed, and manual pressure was held.
Total procedure time: 40 minutes. Estimated blood loss: <50mL. The patient tolerated the procedure well. He was discharged on dabigatran the following day. At four-week follow-up, stent patency was maintained per venous duplex ultrasound, and the patient’s right lower extremity pain and edema had improved. Progress was also seen in a previously non-healing ulceration of the right foot.
He will continue wound care and daily medication and return every six months for repeat imaging.
Conclusion
Use of the novel RevCore system (F) allowed for thorough extraction of in-stent thrombosis and complete lumen restoration post-thrombectomy, with sustained patency and symptom relief at follow-up. The results show promise for improving quality of life in patients who had exhausted their treatment options previously.











