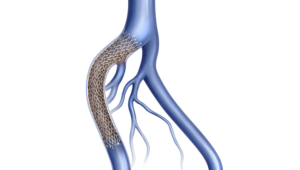
BD recently announced that its Venovo venous stent is back on the U.S. market following a recall last year.
In 2019, the company reported that the Food and Drug Administration (FDA) had granted premarket approval for the Venovo venous stent. In a recall notification dated May 12, 2021, however, BD communicated that the company had expanded a safety notice issued earlier in the year for the Venovo venous stent system to include all sizes and lots within expiry date.
The company details that the Venovo venous stent is a flexible nitinol stent specifically designed to reopen blocked iliac and femoral veins in order to maintain adequate blood flow. The stent is designed with a balance of radial strength, compression resistance and flexibility needed for the treatment of symptomatic post-thrombotic and non-thrombotic iliofemoral lesions. Additionally, the broad stent sizing allows clinicians to treat large diameter veins and long lesion lengths.
“The unique attributes of the Venovo stent make it particularly well-suited to treat iliofemoral occlusive disease,” says Michael Dake, MD, of the University of Arizona, Tucson, Arizona, the principal investigator for the Venovo investigational device exemption (IDE) trial. “Most importantly,” says Dake, “it is purpose-built for application in veins, and engineered to address the special challenges of venous lesions that are very different than those posed by arterial narrowing.”
Dake presented positive one- and two-year results of the VERNACULAR trial of the Venovo stent at meetings in 2018 and 2019, respectively.










