Tag: IDE
Survey of off-label treatment of complex repair pinpoints factors behind underreported...
The potential for cost and time to be key factors in low levels of outcomes data reporting emerged during a national survey of the...
Vascular community has a role to play in ensuring safety of...
Physician-sponsored Investigational Device Exemption (IDE) trials have an important role to play in the continued development and refinement of aortic endovascular devices, attendees of...
Analysis will kickstart conversation on F/BEVAR outcomes
New data presented during Friday's Plenary Session 6 (10–11 a.m. in the West Building, Skyline Ballroom) will shed light on trends in the adoption...
Getinge and Cook Medical enter US commercial distribution agreement for iCast...
Getinge and Cook Medical today announced an exclusive sales and distribution agreement for the iCast covered stent system, which recently received Food and Drug...
Florida IDE trial data support use of physician-modified F/BEVAR over open...
This advertorial is sponsored by Tampa General Hospital.
As Dean Arnaoutakis, MD, describes it, failed endovascular aortic aneurysm repair (EVAR) for abdominal aortic aneurysms (AAAs)...
First patient treated in ZFEN+ fenestrated endovascular graft study
Cook Medical has announced the first patient treated in the clinical study of the Zenith Fenestrated+ endovascular graft (ZFEN+) in the U.S. The procedure was...
Enrollment complete in APEX-AV study of mechanical aspiration system for acute...
Patient enrollment is now complete in the APEX-AV trial evaluating the safety and efficacy of the AlphaVac F1885 (AngioDynamics) multipurpose mechanical aspiration system for...
Enrollment of first patient in Neuroguard carotid stent system trial announced
Contego Medical has announced enrollment of the first patient in the prospective, multicenter PERFORMANCE III trial aimed at further evaluating the safety and effectiveness of the...
Light-activated drug-coated balloon granted FDA approval for clinical study
Alucent Biomedical has announced that the Food and Drug Administration (FDA) granted an investigational device exemption (IDE) for a U.S. clinical study of AlucentNVS,...
‘FDA’s mission to bring safe and effective vascular surgery devices to...
The Vascular and Endovascular Devices Team (VEDT) in the Food and Drug Administration (FDA) Office of Cardiovascular Devices appreciates the Vascular Specialist editorial board's...
FDA approves study of ZFEN+ for treatment of aortic aneurysms
The Food and Drug Administration (FDA) has granted approval for Cook Medical to initiate an investigational device exemption (IDE) study on the Zenith fenestrated+...
Getting medical devices to market: The future might not be now,...
The development of medical devices in the vascular space faces increasing challenges amid moves afoot at the Food and Drug Administration (FDA), according to...
First US patient enrolled in Gore Viafort vascular stent pivotal study
W L Gore & Associates (Gore) has announced that the first U.S. patient has been enrolled in a prospective, randomized, multicenter, single-arm study with...
How a multidisciplinary team meets the complex challenges of fixing failed...
This advertorial is sponsored by Tampa General Hospital.
The Aortic Disease Program at Tampa General Hospital’s Heart & Vascular Institute is no stranger to the...
Selution SLR receives second FDA IDE approval
Selution SLR, MedAlliance’s sirolimus-eluting balloon, has received conditional Food and Drug Administration (FDA) investigational device exemption (IDE) approval to initiate its pivotal clinical trial...
AVeVA study confirms benefit of covered stent placement in graft-vein anastomotic...
A prospective, multicenter study involving the Covera vascular covered stent (BD) has confirmed the benefits of immediate, post-percutaneous transluminal angioplasty (PTA) placement of the...
First patient enrolled in Wrapsody endoprosthesis registry study
Merit Medical has announced the successful enrollment of the first patient in its WRAP registry study in a press release. The study will evaluate...
Venovo venous stent returns to US market after 2021 recall
BD recently announced that its Venovo venous stent is back on the U.S. market following a recall last year.
In 2019, the company reported that the...
Complex TAAA: US ARC IDE registry reports better survival vs. real-world...
Patients treated in Food and Drug Administration (FDA)-approved investigational device exemption (IDE) trials for complex thoracoabdominal aortic aneurysms (TAAAs) in the U.S. Aortic...
Vascular Specialist–February 2022
In this issue:
"Vital signs": Reporting from the Southern Association for Vascular Surgery (SAVS) annual meeting on skyrocketing nursing costs and their contribution to...
The top 10 most popular Vascular Specialist stories in January
January’s top 10 includes “Our forever plague,” a combined editorial by Malachi Sheahan III, MD, our medical editor, on fake news and science, and...
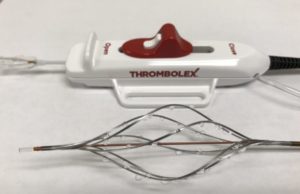
Thrombolex: Investigational RESCUE trial yields ‘exciting’ interim data
The prespecified interim analysis of the first 62 evaluable pulmonary embolism (PE) patients enrolled in the investigational RESCUE trial produced positive results, Thrombolex has...
US IDE Aortic Research Consortium ‘pushes the envelope’ on F/BEVAR
The U.S. investigational device exemption (IDE) Aortic Research Consortium has produced the strongest data so far on the use of fenestrated/branched endovascular aneurysm repair...
Endologix announces completion of enrollment in TORUS 2 study for PAD...
Endologix has completed enrollment in the TORUS 2 investigational device exemption (IDE) clinical study in the U.S., a press release reports.
The TORUS 2 study...
VenoValve: Chronic venous insufficiency device gains brisk double of first US...
Hancock Jaffe Laboratories recently revealed that the United States Patent and Trademark Office (USPTO) issued the first patent covering the company’s VenoValve. The patent is...
Treo IDE primary endpoint results announced
In the wake of approval by the Food and Drug Administration (FDA) of the Treo abdominal aortic stent-graft system for the treatment of patients...
FDA grants Nexus aortic arch stent graft system breakthrough designation
Endospan was recently granted breakthrough device designation from the Food and Drug Administration (FDA) for the Nexus aortic arch stent graft system.
The FDA’s breakthrough...
Aortic neck dilation after FEVAR leads to little clinical impact, new...
MIAMI—Data, gathered in a study on the impact of aortic neck dilation after fenestrated endovascular aortic aneurysm repair (FEVAR), show that the process does...