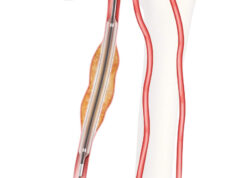
Patients treated with Boston Scientific’s Ranger drug-coated balloon (DCB) sustained “improved primary patency” with fewer reinterventions than those treated with uncoated devices, two-year results from the RANGER II SFA randomized controlled trial have demonstrated, investigators revealed during a late-breaking session at Vascular Interventional Advances (VIVA) 2021 (Las Vegas, Oct. 5–7).
Principal investigator Ravish Sachar, MD, an interventional cardiologist at North Carolina Heart and Vascular at UNC REX Healthcare in Raleigh-Durham, delivered the data, telling attendees that subgroup analyses—covering women, patients with baseline chronic total occlusions [CTOs] and calcified lesions—also suggest “consistent benefit” of DCB treatment vs. standard percutaneous transluminal angioplasty (PTA).
Additionally, the two-year results showed no difference in mortality rate between the groups.
In a press release, Boston Scientific said the data confirmed the safety and efficacy of the Ranger DCB compared to standard PTA for the treatment of patients with peripheral artery disease (PAD) in the superficial femoral artery (SFA) and proximal popliteal artery. The new findings follow the May 2021 publication of the one-year results of RANGER II SFA in JACC: Cardiovascular Interventions.
Sachar and colleagues found that the Ranger DCB exhibited a higher primary patency rate of 84% compared to 71.4% in patients treated with standard PTA (p=0.0129). Additionally, the subgroup analyses found consistent benefit with greater long-term patency in patients with more complex lesions treated with the Ranger DCB, exhibiting an 89.1% vs. 72.4% primary patency rate in the moderate to severe calcium subgroup (p=0.0052) and a 76.6% compared to a 58.6% primary patency rate in patients with CTOs (p=0.1038).
“These two-year data demonstrate a sustained, high rate of efficacy—including in patients with more complex lesion subtypes, yet another proof point for physicians to consider when determining the best individualized treatment option for their patients with PAD,” said Sachar.
The Ranger DCB also demonstrated a significant reduction in reinterventions at two years, with a freedom from target lesion revascularization (TLR) rate of 87.4% vs. 79.5% observed with standard PTA (p=0.0316).i Additionally, there was no significant difference in all-cause mortality: 5.7% for Ranger DCB patients and 3.2% in those treated with standard PTA (p=0.4218). Sachar added that there was no reintervention disadvantage based on gender.