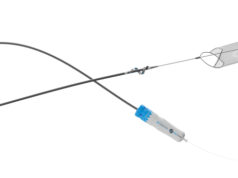
Surmodics has announced that J. Michael Bacharach, MD, a vascular interventionalist/cardiologist at North Central Heart, a division of Avera Heart Hospital in Sioux Falls, South Dakota, successfully treated the first patient with the company’s Pounce thrombectomy system.
The procedure involved a patient presenting with increasing pain in the left leg and was diagnosed with an occluded superficial femoral artery (SFA). Three passes of the Pounce device removed 30cm of subacute thrombus and blood flow was successfully restored. Eight additional cases have since been performed in the U.S. with positive outcomes reported following each procedure.
“Despite the challenge presented by this particular case, I could not have asked for anything more. The device’s performance was really amazing,” said Bacharach. “I was able to safely and effectively achieve a positive outcome for this patient with minimal blood loss and without the use of thrombolytics. I’m very excited about the results of this case. I’m a believer in the Pounce system.”
The Pounce Thrombectomy System, which received Food and Drug Administration (FDA) 510(k) clearance in September 2020, is a mechanical thrombectomy device intended for the non-surgical removal of thrombi and emboli from the peripheral arterial vasculature.
Depending on the age and magnitude of the occlusion and the viability of the threatened limb, existing treatments for this condition may include catheter directed thrombolysis, surgical embolectomy, and/or percutaneous mechanical thrombectomy. In cases in which the occlusion has caused irreversible damage to the limb, acute limb ischemia can result in the amputation of a lower extremity.
Gary Ansel, MD, founder and former chief medical officer of Embolitech—from which Surmodics acquired the technology and intellectual property behind the Pounce thrombectomy system—said: “I am delighted that the Surmodics team is providing peripheral interventionalists with this innovative tool for treating arterial thrombotic occlusions.
“I also congratulate the company on its continued efforts to advance therapies for patients dealing with arterial thrombosis and peripheral arterial disease while focusing on ease of use and efficiency for the physicians treating these patients.”