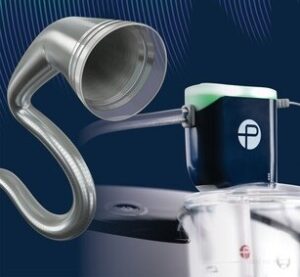
Penumbra have announced the Food and Drug Administration (FDA) clearance and launch of its Lightning Flash mechanical thrombectomy system.
“Lightning Flash features Penumbra’s novel Lightning intelligent aspiration technology, now with dual clot detection algorithms,” the company notes in a press release, adding that the system is designed to quickly remove large blood clots in the body, including venous thrombus and pulmonary emboli (PE).
“Penumbra’s Lightning Flash gives physicians a highly torqueable, larger catheter that is designed to remove a large clot burden in the pulmonary arteries or deep venous system more efficiently while maintaining an excellent safety profile because of Lightning’s computer-aided algorithms,” said James F. Benenati, MD, chief medical officer at Penumbra. “We believe that patients can have improved outcomes with this new technology because of the exceptional trackability and unique ability to distinguish flowing blood from clot.”
The company details that Lightning Flash, powered by the Penumbra Enging, uses both pressure and flow-based algorithms to detect blood clot and blood flow. The catheter is made with MaxID hypotube technology, allowing an inner diameter similar to large-bore catheters while maintaining a lower profile and a soft, atraumatic tip designed to help navigate the complex and delicate anatomy of the body. When used together, the device is intended to help remove blood clots quickly while minimizing potential blood loss.
Lightning Flash is part of Penumbra’s Indigo system with Lightning portfolio.











