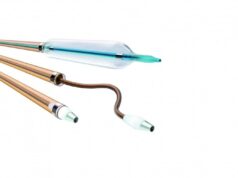
Royal Philips has announced the final, five-year results of two randomized controlled trials (RCTs) that show no difference in all-cause mortality between patients treated with the Stellarex drug-coated balloon (DCB) and those treated with percutaneous transluminal angioplasty (PTA), the current standard of care.
The studies showed no difference in mortality between the Stellarex DCB and PTA at every 12-month endpoint over the course of the study. Results from the two RCTs were presented at the ongoing LINC 2021 (Leipzig Interventional Course).
The ILLUMENATE EU RCT and ILLUMENATE Pivotal RCT together comprised approximately 600 patients in Europe and the United States. After five years, the ILLUMENATE EU RCT showed 19.3% mortality among patients treated with the Stellarex DCB compared to 19.4% mortality for those treated with PTA.
The five-year results for the ILLUMENATE Pivotal study also show no statistically significant difference among patients treated with the Stellarex DCB (21.2%) compared to those treated with PTA (20.2%). Both studies had a high vital status follow-up compliance, with the status of over 90% of patients known.
“The five-year final results show favorable overall safety of the Stellarex low-dose paclitaxel DCB, and are applicable within an extensive patient cohort,” said Marianne Brodmann, MD, of the Medical University of Graz in Graz, Austria, primary investigator for both trials and a paid consultant to Philips. “Every year, the Stellarex program has demonstrated low mortality rates with no difference in rates between the two patient cohorts.”