In Physics of Fluids, by AIP Publishing, researchers from the Indian Institute of Technology (BHU) Varanasi and Indian Institute of Technology Kanpur made a computational model of the cardiovascular system in order to predict early abdominal aortic aneurysm (AAA) rupture and monitor patients’ blood vessel conditions.
The team investigated the effect of realistic, patient-specific AAA shapes on the hemodynamics of pulsatile Newtonian fluids in an aortofemoral artery under normal and diseased conditions.
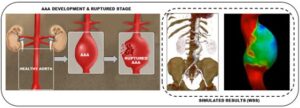
An AAA causes the wall of a person’s aorta, the largest artery in the body, to weaken and bulge outward. If left untreated, it can continue to grow and eventually rupture, which can lead to life-threatening bleeding.
According to some estimates, up to 80% of patients who experience a ruptured AAA will die before they reach the hospital or during surgery. But early intervention can prevent rupture, improve outcomes, and avoid death.
Predicting the risk of AAA rupture involves a combination of imaging studies, such as ultrasound, computed tomography (CT) scans, and magnetic resonance imaging (MRI), and hemodynamics, as well as clinical factors such as age, sex, smoking history, and family history of AAA.
“If an AAA is detected early, treatment options such as surgical repair or endovascular stent grafting are available to prevent rupture,” said the authors. “These treatments are both effective at reducing the risk of rupture and improving survival rates.”
Using image-based computational blood dynamics, the researchers mimicked specific health conditions and investigated various hemodynamic parameters. Their patient-specific geometric models of a human aortofemoral artery were constructed from three-dimensional (3D) medical imaging data. To solve the blood flow governing equations under the pulsating conditions caused by the heart’s beating, they used finite element-based simulations.
The team found that aneurysm size alters the blood flow velocity distribution. In addition, flow separation occurs during systolic deceleration, and the vortex begins to travel in the aneurysm sac. Among other complex dynamics, this may influence the blood circulation of lower extremities.
“In the future, such computational work will help in development of digital twins of the cardiovascular system,” said Sumit Kumar, PhD, lead author of the study.
Digital twins are virtual patient representations that receive real-time updates on a variety of data variables and help doctors better forecast disease and choose the best course of therapy.












