
This advertorial is sponsored by Shockwave Medical.
Mazin Foteh, MD, contrasts the benefits of Shockwave Medical’s new Shockwave L6 Peripheral Intravascular Lithotripsy Catheter alongside those of its sister device, the Shockwave M5+ Peripheral Intravascular Lithotripsy Catheter.
The Shockwave L6 device is a purpose-built IVL catheter, perfectly designed for complex iliac occlusive disease. Calcified plaque blockages in this region are distinctly different from lesions elsewhere in the arterial system. These are much larger blood vessels, which are often thick and heterogeneously calcified. Although the Shockwave M5+ IVL catheter was instrumental in introducing this technology to an underserved space, in some scenarios it lacked the size and energy profile to compete with this degree of calcium. The most important feature of the Shockwave L6 catheter is its larger diameter offerings (8, 9, 10 and 12mm).
Today, we can appropriately match the Shockwave balloon catheter to the vessel and optimize outcomes. Secondly, the compact emitter placement is instrumental in providing a uniform energy profile across the length of the balloon. There are a total of three paired emitters, which fire in groupings of two. This translates to consistent sonic output without diminishment at the ends.
The Shockwave M5+, on the other hand, delivers peak pressure at the center electrode. In practice this equates to improved case efficiency, as we are no longer trying to center the electrode at the most dense area of plaque. Finally, all of the Shockwave L6 catheters can be delivered over an 0.018” wire and in 7/8F sizes. Tracking over a larger wire removes resistance and sets a platform for additional therapies such as stents.
To date, the endovascular treatments for calcified iliac plaques have centered on the “crack and pave” technique, which harnesses barotrauma as the mechanism of action. The Shockwave L6 catheter allows a rethinking of this method, now relying purely on low-pressure inflations and the delivery of sonic pressure waves to the hardened calcium.
This results in a much gentler, worry-free technique, causing calcified lesions to yield in a manner that is without the risk of rupture. Additionally, if stenting is necessary, we can rest assured that optimal luminal gain can be achieved. This particular arterial bed is very frequently laden with calcium deposits, therefore the need for new techniques is present.

The Shockwave L6 catheter is the first technology to address this need and may one day challenge the notion that all iliac blockages require a stent. My practice is centered around large-bore devices for endovascular aneurysm repair (EVAR), thoracic EVAR (TEVAR), and fenestrated EVAR (FEVAR). Iliac stenosis and poor access are quite common in this population.
Prior to the Shockwave L6, we were forced to create a runway for delivery. This meant advanced and expensive techniques such as endovascular and open surgical conduits. These procedures can add cost, but also morbidity. I can now successfully treat most iliac lesions in this setting with the Shockwave L6 alone.
The Shockwave M5+ was an excellent precursor to the Shockwave L6 catheter. Its longer length certainly provided a nice platform to treat multi-segment disease, especially in the external iliac as well as the fem-pop distribution. However, its energy profile differs greatly from the Shockwave L6, with the maximal energy coming from the center electrode. This feature affects the way the balloon is used, generally requiring significant overlap of the inflations and sometimes even trying to center the middle emitter on the peak plaque.
Furthermore, it is somewhat limited in its applicability in larger vessels, simply based on the balloon size. I find that for medium-sized vessels with moderate calcium burden that this balloon is favored. Whereas for focal, dense plaque in larger vessels, the Shockwave L6 is the catheter of choice. My experience with this system has taught me that appropriate balloon sizing is the single-most important predictor of a successful outcome.
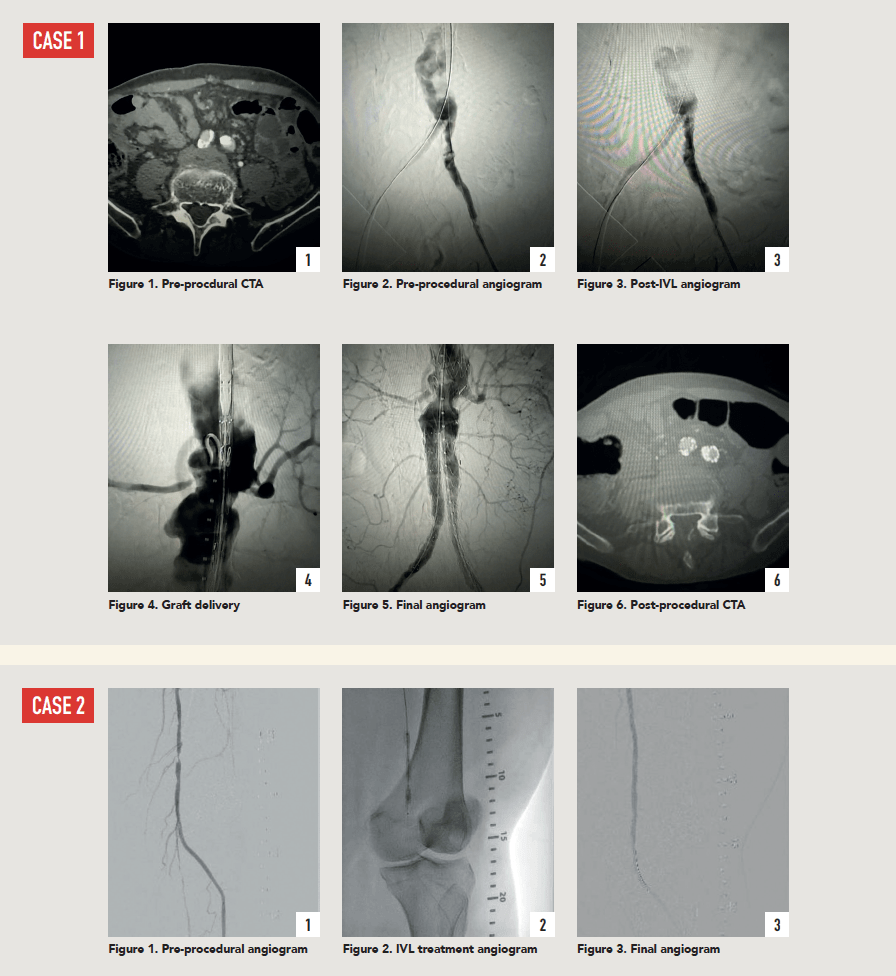
Case 1: The Shockwave L6 device in the setting of FEVAR and complex iliac stenosis
An 82-year-old man with a history of coronary artery disease (CAD), hypertension, hyperlipidemia, and peripheral arterial disease (PAD) presents with rest pain as well as a large pararenal abdominal aortic aneurysm (AAA).
His computed tomography angiography (CTA) revealed very dense and highly diseased iliac vessels with calcium deposition along most of the bilateral common iliac distribution.
The minimum luminal diameter (MLD) of the common iliac arteries measured roughly 4.5mm; however, the healthy reference vessel diameter (RVD) was 9.5mm. He would require a fenestrated repair of the AAA along with complex iliac stenosis treatment, and extensive bilateral common femoral endarterectomies.
Further, we were concerned we would not be able to advance the 19F fenestrated graft. The endograft was prepared, and then both femoral arteries were exposed. We attempted delivery of the endograft via the right common iliac access. This failed. We then attempted delivery on the contralateral side. Despite multiple attempts, we made little progress. A 10x30mm Shockwave L6 catheter was selected, and a total of 150 pulses were delivered to each iliac artery at just 4atm. This not only allowed for the delivery of our FEVAR graft but also the full unrestricted expansion of the endograft limbs in the common iliac arteries.
At the completion of the AAA, we took extra time to perform endarterectomies of the deep femoral arteries. His completion CT scan revealed a good seal of the AAA, patency of the renal fenestrations and unrestricted flow to the bilateral femoral arteries.
Case 2: The Shockwave M5+ tackles 90% stenosis of the distal SFA
The patient is a 75-year-old man with a history of hypertension, hyperlipidemia, CAD, chronic kidney disease and diabetes mellitus, who presents with a non-healing wound of the right lower extremity for three months.
He had undergone diagnostic work—his ankle-brachial index (ABI) was found to be 0.73 with a toe pressure of 50mmhg, and a duplex ultrasound revealed a densely calcified superficial femoral artery (SFA) stenosis of 90%.
He was taken to the cath lab and underwent a right lower-extremity arteriogram. This confirmed a 90% stenosis of the distal SFA, with normal three-vessel run off.
Due to the heavy calcium, we opted for a Shockwave M5+ 6x60mm and delivered 200 pulses to the lesion. There was no appreciable residual stenosis at the completion of the procedure, and his post-procedure ABI improved to 1.
This is a great example of how IVL can be used as a standalone therapy without the need for adjunctive treatment.
Mazin Foteh is director of aortic therapy and innovation and co-director of the Aortic Clinic at Baylor Scott & White Health in Plano, Texas. He is a paid consultant to Shockwave Medical. The views expressed are those of the author and not necessarily those of Shockwave Medical.
 Shockwave M5+, Shockwave M5, Shockwave S4 and Shockwave L6 Safety Information In the United States: Rx only. Indications for Use—The Shockwave Medical Intravascular Lithotripsy (IVL) System is intended for lithotripsy-enhanced balloon dilatation of lesions, including calcified lesions, in the peripheral vasculature, including the iliac, femoral, ilio-femoral, popliteal, infra-popliteal, and renal arteries. Not for use in the coronary or cerebral vasculature.
Shockwave M5+, Shockwave M5, Shockwave S4 and Shockwave L6 Safety Information In the United States: Rx only. Indications for Use—The Shockwave Medical Intravascular Lithotripsy (IVL) System is intended for lithotripsy-enhanced balloon dilatation of lesions, including calcified lesions, in the peripheral vasculature, including the iliac, femoral, ilio-femoral, popliteal, infra-popliteal, and renal arteries. Not for use in the coronary or cerebral vasculature.
Contraindications—Do not use if unable to pass 0.014” (M5, M5+, S4) or 0.018” (L6) guidewire across the lesion-Not intended for treatment of in-stent restenosis or in coronary, carotid, or cerebrovascular arteries.
Warnings—Only to be used by physicians who are familiar with interventional vascular procedures—Physicians must be trained prior to use of the device—Use the generator in accordance with recommended settings as stated in the Operator’s Manual.
Precautions—use only the recommended balloon inflation medium—Appropriate anticoagulant therapy should be administered by the physician—Decision regarding use of distal protection should be made based on physician assessment of treatment lesion morphology.
Adverse effects–Possible adverse effects consistent with standard angioplasty include–Access site complications –Allergy to contrast or blood thinner– Arterial bypass surgery—Bleeding complications— Death—Fracture of guidewire or device—Hypertension/Hypotension—Infection/sepsis—Placement of a stent—renal failure—Shock/pulmonary edema—target vessel stenosis or occlusion—Vascular complications. Risks unique to the device and its use—Allergy to catheter material(s)— Device malfunction or failure—Excess heat at target site.
Prior to use, please reference the Instructions for Use for more information on indications, contraindications, warnings, precautions and adverse events. www.shockwavemedical.com. SPL-68917 Rev. A.











