Tag: BTK
FDA grants R3 Vascular IDE approval for ELITE-BTK pivotal trial of...
R3 Vascular today announced that the Food and Drug Administration (FDA) has granted investigational device exemption (IDE) approval to initiate its ELITE-BTK pivotal trial...
FDA Breakthrough Device designation granted for DynamX BTK for use in...
Elixir Medical has announced Breakthrough Device designation by the Food and Drug Administration (FDA) for its novel DynamX BTK System, an implant for use...
PRELUDE-BTK subanalysis highlights superior lumen gain, greater volumetric blood flow using...
Cagent Vascular has announced the results of a comparative subanalysis of the PRELUDE-below-the-knee (BTK) study versus plain balloon angioplasty.
The study was led by Marianne...
Revascularization strategy for BTK intervention should hinge on diagnostic modality, LINC...
New data provide “another argument” for the use of intravascular ultrasound (IVUS) in below-the-knee (BTK) interventions. This is according to Michael Lichtenberg, MD, from Arnsberg...
Cook Medical receives FDA breakthrough designation for new drug-eluting stent
Cook Medical has received Breakthrough Device designation from the Food and Drug Administration (FDA) on a new drug-eluting stent (DES) for below the knee...
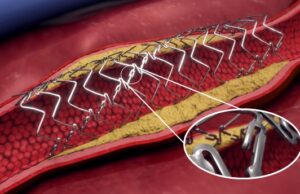
Philips announces positive two-year data from TOBA II BTK clinical trial
Royal Philips recently announced positive two-year results from the TOBA (Tack optimized balloon angioplasty) II below-the-knee (BTK) clinical trial.
The data show the Philips Tack endovascular...
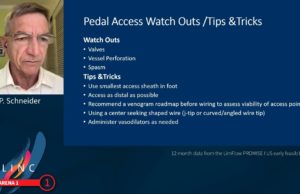
Innovation: ISET audience hears what’s on the horizon for CLTI patients
“I am going to give you reasons to be excited about your critical limb practice,” Peter Schneider, MD, professor of vascular and endovascular surgery...
MicroStent to treat PAD below the knee receives FDA breakthrough device...
Micro Medical Solutions (MMS) recently revealed Food and Drug Administration (FDA) breakthrough device designation for its MicroStent vascular stent. This novel technology is designed...
FDA panel recommends against premarket approval of Lutonix 014 DCB for...
A Food and Drug Administration (FDA) advisory panel has recommended against premarket approval of BD’s Lutonix 014 drug-coated balloon (DCB) for use in below-the-knee (BTK) arteries....
Data show potential of Limflow system to improve outcomes for no-option...
“Deep vein arterialization is safe and technically feasible,” Peter Schneider, MD, a professor in the division vascular and endovascular surgery at the University of...
First US commercial use of Tack Endovascular System in BTK arteries...
Intact Vascular has announced the first commercial use of its Tack Endovascular System (4F) in multiple sites across the United States.
Notably the first Food...