 StentIt has announced the successful implantation of its Resorbable Fibrillated Scaffold (RFS). As part of the VITAL-IT 1 study, patients with chronic limb-threatening ischemia (CLTI) below the knee have successfully been treated using the RFS implant. In this clinical procedure, revascularization was achieved using an endovascular approach to restore blood flow to the foot.
StentIt has announced the successful implantation of its Resorbable Fibrillated Scaffold (RFS). As part of the VITAL-IT 1 study, patients with chronic limb-threatening ischemia (CLTI) below the knee have successfully been treated using the RFS implant. In this clinical procedure, revascularization was achieved using an endovascular approach to restore blood flow to the foot.
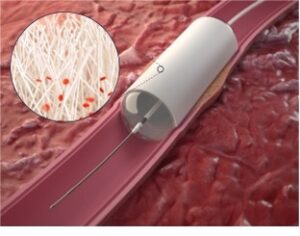
StentIt’s RFS device is a bioresorbable stent built from microfibers, providing structural support to instantly open, and facilitate the reconstruction of the artery. Due to the porous design of the implant, patient’s own cells infiltrate into the mesh, triggering the formation of new vascular tissue. While the artery is being reconstructed from the inside-out, the synthetic implant gradually resorbs and ultimately disappears over time.
VITAL-IT 1 (NCT07006467) is a prospective, non-randomized feasibility study, designed to evaluate StentIt’s RFS device in up to 10 patients with below-the-knee CLTI. This single-centre study is being conducted at the Medical University of Graz, Austria. All study patients will be monitored for 24 months.
“This first-in-human clinical study will provide an important indication on the translational potential of this new technology in CLTI patients,” said Marianne Brodmann, MD, from Medical University of Graz in Graz, Austria. “This device combines key attributes of temporary structural support with regenerative properties, which could minimize the need for reinterventions.”











