Tag: VOYAGER PAD trial
Digging deep into DOACs for PAD post-revascularization in fight to stave...
This advertorial is sponsored by ACHL.
“It’s really sad—but it’s true: In the PAD space, we’re really behind. We’re behind in our understanding of the...
FDA approves expanded PAD indication for rivaroxaban plus aspirin
The Janssen Pharmaceutical Companies of Johnson & Johnson today announced that the Food and Drug Administration (FDA) has approved an expanded peripheral arterial disease...
VOYAGER PAD: Rivaroxaban plus aspirin should be considered after lower-extremity bypass...
“A strategy of adding rivaroxaban 2.5mg twice daily to aspirin should be considered after lower extremity bypass regardless of conduit type,” concluded Nicholas Govsyeyev,...
The top 10 most popular Vascular Specialist stories in May
A pointed letter aimed at electronic health record (EHR) behemoth Epic, an argument in favor of endovascular treatment of ruptured abdominal aortic aneurysms (rAAAs)...
ACC.21: VOYAGER PAD shows rivaroxaban ‘significantly’ reduced total ischemic events in...
Rivaroxaban combined with low-dose aspirin led to a significant reduction in the occurrence of total ischemic events in patients with symptomatic peripheral arterial disease...
VOYAGER PAD: Rivaroxaban plus aspirin versus aspirin alone among CLTI patients...
In one of the latest sub-analyses from the VOYAGER PAD trial, investigators found that rivaroxaban plus aspirin versus aspirin alone in the high-risk population...
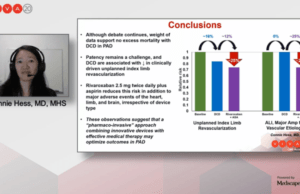
Latest VOYAGER PAD analysis finds no mortality and improved limb outcomes...
Given the absence of a safety signal in data from the VOYAGER PAD trial, a new analysis examined the potential benefit of drug-coated device...
Janssen applies to FDA for new indication to expand use of...
The Janssen Pharmaceutical Companies of Johnson & Johnson announced today it has submitted a supplemental New Drug Application (sNDA) to the Food and Drug...
TCT 2020 late-breaking trials session features new data on drug-coated devices
Findings from a series of late-breaking trials in the endovascular field were delivered recently at TCT Connect (Oct. 14–18), the 32nd annual scientific symposium...
Clopidogrel exposure along with aspirin and rivaroxaban should be ‘minimized or...
“More bleeding with background clopidogrel, even if not severe by adjudication, may be associated with broad consequences, including discontinuation of therapies. In the absence...