Tag: VIVA 2022
VQI study leader highlights virtue of multispecialty gathering of best longitudinal...
Daniel Bertges, MD, talks to Vascular Specialist about recent findings from his work investigating the IN.PACT Admiral drug-coated balloon (DCB). He related how they...
Paclitaxel DCB performs well in ‘challenging anatomy’ of femoropopliteal in-stent restenosis,...
The IN.PACT Admiral drug-coated balloon (DCB) shows promising results in treating femoropopliteal in-stent restenosis (ISR) in a Society for Vascular Surgery (SVS) Vascular Quality...
Vascular Specialist–November 2022
In this issue:
First data from BEST-CLI are revealed at the American Heart Association (AHA) Scientific Sessions (p. 1 and 12–13)
The Society...
Disrupt PAD III observational study results confirm safety and effectiveness of...
The final 1,373-patient cohort analysis from the Disrupt PAD III observational study (OS) demonstrates consistent intravascular lithotripsy (IVL; Shockwave Medical) outcomes in complex and...
SurVeil DCB demonstrates sustained durability of safety, efficacy endpoints in TRANSCEND
In the TRANSCEND clinical trial, the SurVeil drug-coated balloon (DCB; Surmodics) demonstrated "excellent efficacy and safety" out to 24-month follow-up. This is according to...
New analysis: Four multicenter trials support primary use of drug-coated balloons...
A patient-level, propensity-adjusted comparison of three-year results from a quartet of prospective, multicenter trials support the primary use of drug-coated balloons (DCBs) versus bare...
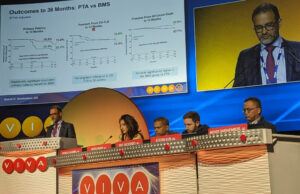
‘Excellent’ three-year results presented for the BioMimics 3D stent in longer...
At the 2022 Vascular Interventional Advances (VIVA) meeting (Oct. 31–3 Nov. 3), Michael Lichtenberg, MD, from Arnsberg Clinic, Arnsberg, Germany, reported a subgroup analysis...