Tag: US FDA
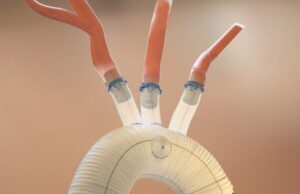
Aquedeon Medical receives FDA approval to expand clinical trial of Duett...
The Food and Drug Administration (FDA) has approved the expansion of Aquedeon Medical's investigational device exemption (IDE) clinical trial evaluating the Duett vascular graft system,...
FDA announces plans to address medical device shortage risks
The Food and Drug Administration (FDA) has issued a statement outlining measures to enhance protections against medical device shortages.
According to Michelle Tarver, director...
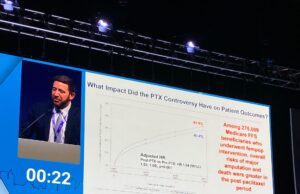
Paclitaxel controversy: Yes, device restrictions did cause harm
“Unfortunately, we are doing worse for our patients today,” were the sobering thoughts of Eric Secemsky (Boston, United States) during a late-breaking presentation in...
TCT 2023: First presentation of complete patient-level dataset on paclitaxel and...
Data from a patient-level meta-analysis—a factor in the US Food and Drug Administration’s (FDA) recent change of position on the use of paclitaxel-coated devices...
Swiss parliament votes to accept FDA-approved medical devices
The Swiss Federal Assembly has voted in favor of accepting medical devices with the Food and Drug Administration (FDA) marketing authorization in Switzerland.
A motion...
FDA advisory panel issues recommendations on lifelong surveillance for EVAR
The Food and Drug Administration (FDA) recently announced it has issued a letter to healthcare providers emphasizing the importance of lifelong surveillance, including imaging,...