Tag: SWEDEPAD
Meta-analysis: Drug-coated devices show comparable aggregate outcomes to non-drug-coated devices in...
Drug-coated balloon (DCB) angioplasty appears to offer similar benefits as plain old balloon angioplasty (POBA), with or without stenting, for patients with chronic limb-threatening...
‘We need to look at the totality of evidence’: VIVA panelists...
A focused session at the 2025 Vascular Interventional Advances (VIVA) conference (2–5 November, Las Vegas, USA) homed in on the latest findings from the...
SWEDEPAD re-opens paclitaxel safety discussion, finds drug-coated devices do not improve...
Drug-coated balloons and stents were not associated with reduced risk of amputation or improved quality of life compared with uncoated devices in the SWEDEPAD...
‘Proceed cautiously in use of DCBs in CLTI patients with advanced...
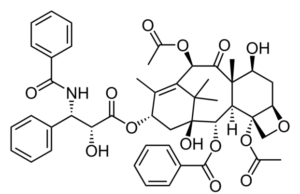
The last several years have seen ongoing controversy and debate about the risks and benefits of paclitaxel-coated devices (PCDs) for patients with peripheral arterial...
FDA representatives respond to SWEDEPAD interim analysis, highlight need for continued...
Representatives from the Food and Drug Administration (FDA) referenced "important and reassuring" results from the recent interim analysis of the SWEDEPAD clinical trial in which...
SWEDEPAD unplanned interim analysis shows no difference in all-cause mortality for...
An unplanned interim analysis of the registry-based SWEDEPAD clinical trial, in which patients with peripheral arterial disease (PAD) received treatment with paclitaxel-coated (drug-coated balloons...